| Identification | More | [Name]
Erlotinib | [CAS]
183321-74-6 | [Synonyms]
4-[(3-ethynylphenyl)amino]-6,7-bis(2-methoxyethoxy)quinazoline
6,7-bis(2-methoxyethoxy)-4-(3-ethynylanilino)quinazoline hydrochloride
ERLOTINIB
ERLOTINIB HCL
ERLOTINIB HCL SALT
erlotinib hydrochloride
n-(3-ethynylphenyl)[6,7-bis(2-methoxyethoxy)quinazolin-4-yl]amine
n-(3-ethynylphenyl)[6,7-bis(2-methoxyethoxy)quinazolin-4-yl]amine hydrochloride
TARCEVA
Erlotinib Hydrochloirde
4-Quinazolinamine, N-(3-ethynylphenyl)-6,7-bis(2-methoxyethoxy)-
Tarceva
ERLOTININ
Erlortinib
N-(3-ethynylphenyl)-6,7-bis(2-methoxyethoxy)-4-quinazolinamine
| [EINECS(EC#)]
689-196-2 | [Molecular Formula]
C22H23N3O4 | [MDL Number]
MFCD07781272 | [Molecular Weight]
393.44 | [MOL File]
183321-74-6.mol |
| Chemical Properties | Back Directory | [Melting point ]
159-160 °C | [Boiling point ]
553.6±50.0 °C(Predicted) | [density ]
1.24 | [storage temp. ]
Keep in dark place,Sealed in dry,Store in freezer, under -20°C | [solubility ]
Chloroform (Slightly), DMSO (Slightly), Methanol (Slightly) | [form ]
Solid | [pka]
5.32±0.30(Predicted) | [color ]
White | [CAS DataBase Reference]
183321-74-6(CAS DataBase Reference) |
| Hazard Information | Back Directory | [Description]
Erlotinib is a tyrosine kinase inhibitor which acts on the epidermal growth factor receptor (EGFR), inhibiting EGFR-associated kinase activity (IC50 = 2.5 μM).1,2 This inhibits tumor growth in human head and neck carcinoma HN5 tumor xenografts in mice with an ED50 value of 9 mg/kg.1 Erlotinib also suppresses cyclin-dependent kinase 2 (Cdk2) activity in breast cancer cells (IC50 = 4.6 μM) and JAK2 mutant JAK2V617F positive hematopoietic progenitor cells (IC50 = 5 μM), which is associated with polycythemia vera, idiopathic myelofibrosis, and essential thrombocythemia.3,4 Formulations containing erlotinib have been used to treat certain forms of cancer, including non-small cell lung cancer.5,6 | [Uses]
A tyrosine kinase inhibitor | [Uses]
antineoplastic;tyrosine kinase inhibitor | [Uses]
Erlotinib HCl is an HER1/EGFR inhibitor with IC50 of 2 nM. | [Definition]
ChEBI: A quinazoline compound having a (3-ethynylphenyl)amino group at the 4-position and two 2-methoxyethoxy groups at the 6- and 7-positions. | [Brand name]
Tarceva
(OSI). | [General Description]
Class: receptor tyrosine kinase
Treatment: NSCLC
Oral bioavailability = 60%
Elimination half-life = 36 h
Protein binding = 93% | [Clinical Use]
ErlotinibTreatment of locally advanced or metastatic non�small cell lung cancer after failure of at least 1 other
regimePancreatic cancer | [Side effects]
- Burning, tingling, numbness or pain in the hands, arms, feet, or legs.
- cough or hoarseness.
- diarrhea (severe)
- difficult or labored breathing.
- fever or chills.
- rash (severe)
- sensation of pins and needles.
- stabbing chest pain.
| [Synthesis]
The synthesis of Erlotinib is as follows:
N-dimethylformamidine, 0.72 g (6.15 mmol) of N '- [2-cyano-4,5-bis (2-methoxyethoxy) phenyl] ) Of 3-aminophenylacetylene and 8 mL of acetic acid were reacted in a 50 mL reaction flask at 125 ° C for 1 hour and cooled to room temperature.20 mL of ice water was added to the mixture, the pH was adjusted to 10 with aqueous ammonia, and the mixture was stirred for 1 hour, suction filtered and the filter cake washed with water until neutral.The filter cake was dried to obtain 2.15 g of erlotinib in a yield of 91.5%.
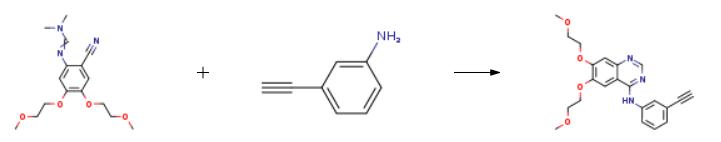 | [Drug interactions]
Potentially hazardous interactions with other drugs
Analgesics: increased risk of bleeding with NSAIDs.Antacids: concentration possibly reduced by
antacids, give at least 4 hours before or 2 hours after
erlotinib. Anticoagulants: increased risk of bleeding with
coumarinsAntipsychotics: avoid concomitant use with
clozapine, increased risk of agranulocytosis.Antivirals: avoid with boceprevir.Ulcer-healing drugs: avoid with cimetidine,
esomeprazole, famotidine, lansoprazole, nizatidine,
pantoprazole and rabeprazole; concentration reduced
by ranitidine, give at least 2 hours before or 10
hours after ranitidine; concentration reduced by
omeprazole - avoid. | [Metabolism]
Erlotinib is metabolised mainly by the cytochrome P450
isoenzyme CYP3A4, and to a lesser extent by CYP1A2.
Extrahepatic metabolism by CYP3A4 in intestine,
CYP1A1 in lung, and 1B1 in tumour tissue potentially
contribute to the metabolic clearance of erlotinib.Metabolic pathways include demethylation, to metabolites
OSI-420 and OSI-413, oxidation, and aromatic
hydroxylation. The metabolites OSI-420 and OSI-413
have comparable potency to erlotinib in non-clinical in
vitro assays and in vivo tumour models. They are present
in plasma at levels that are <10 % of erlotinib and display
similar pharmacokinetics as erlotinib. Erlotinib is excreted
predominantly as metabolites via the faeces (>90%) with
renal elimination accounting for only a small amount
(approximately 9%) of an oral dose. Less than 2% of the
orally administered dose is excreted as parent substance |
| Questions And Answer | Back Directory | [Binding Mode]
Erlotinib binds to the ATP-binding pocket of active EGFR as a type I inhibitor. At the same time, it behaves as a type I1/2 inhibitor in binding to the inactive DFG-in and C helix-out conformation. In both co-crystal structures, the quinazoline N1 hydrogen bonds with the amide nitrogen of Met769 in the hinge region, while the N3 is interacting indirectly with the side chain hydroxyl of Thr766 via a bound water molecule.
| [Molecular targeted therapy]
The small molecule compound, Erlotinib is a receptor tyrosine kinase inhibitor (EGFR antagonist) and belongs to molecular targeted therapy Drugs. It can inhibit the phosphorylation reaction through competing with adenosine triphosphate to bind to the catalytic region of the receptor tyrosine kinase, thereby blocking the down-proliferation signaling and inhibiting the activity of the tumor cell ligand-dependent HER-1/EGFR, thus achieving the inhibition of the proliferation of tumor cells. Clinically, it is mainly used in the treatment of incurable locally advanced or metastatic non-small cell lung cancer (NSCLC) and being used in combination with gemcitabine for first-line treatment of locally advanced unresectable or metastatic pancreatic cancer.
In November 2004, the product was first approved in the United States for the treatment of local advanced or metastatic non-small cell lung cancer (NSCLC) which has been undergone at least one time of chemotherapy failure.
In 2005, the results of a Phase III trial of the NCI Canadian Clinical Trials Group suggested that gemcitabine, in combination with erlotinib (EGFR blocker) can increase the median survival of patients with advanced pancreatic cancer from 5.9 months to 6.4 months and 1-year survival from 17% to 24%.
In November 2005, Genentech and OSI jointly announced that the US Food and Drug Administration (FDA) had approved the combination of erlotinib (Tarceva) and gemcitabine as first-line treatment for advanced pancreatic cancer. | [Gefitinib]
Gefitinib is an aniline quinazoline derivative and is also a selective epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor that inhibits the growth, metastasis and angiogenesis of tumors and increases tumor cell apoptosis;
Gefitinib is a kind of highly-specific anti-tumor targeted therapy drugs developed by the United Kingdom AstraZeneca, being the first molecular targeted drug for the treatment of non-small cell lung cancer. It takes effects through selectively inhibiting the signal transduction pathway of the epidermal growth factor receptor tyrosine kinase (EGFR-TK). Epidermal growth factor (EGF) is a polypeptide with a relative molecular mass of 6.45 × 103 that binds to the epidermal growth factor receptor (EGFR) on the target cell membrane to produces biological effects. EGFR is a tyrosine kinase (TK) receptor that, when conjugated to EGF, being capable of promoting the TK activation in the receptor, leading to auto-phosphorylation of the receptor tyrosine residue, providing a continuous signal splitting into the cell, further causing cell proliferation and differentiation. EGFR is abundant in human tissues and highly expressed in malignant tumors. Gefitinib, through interfering with the EGFR signaling transduction pathways on the cell surface, inhibits the tumor growth, metastasis and angiogenesis and induces tumor cell apoptosis.
In August 2002, gefitinib, as a first-line treatment of non-small cell lung cancer drugs, was first listed in Japan under the trade name of Iressa.
In May 2003, the US Food and Drug Administration approved gefitinib as the three-line monotherapy drug for patients with advanced non-small cell lung cancer that can’t be cured by platinum-based anti-cancer drugs and docetaxel chemotherapy. Now, it has been also approved by Australia, Japan, Argentina, Singapore and South Korea and other countries for the treatment of advanced non-small cell lung cancer.
On February 28, 2005, the Chinese Food and Drug Administration approved gefitinib for the treatment of locally advanced or metastatic non-small cell lung cancer (NSCLC) previously subjecting to chemotherapy treatment. It has not yet been approved as a first-line treatment for advanced NSCLC.
On July 1, 2009, the European Union Drug Administration has formally approved gefitinib for the first, second and third line treatment of adult EGFR mutations locally advanced or metastatic non-small cell lung cancer. | [Dosage and Usage]
Non-small cell lung cancer: 150mg/d; administrate at least1h before meals or 2h after meal; continue taking the drug until the disease get progression or the emergence of intolerable toxicity response when the patients should withdraw the drug.
Pancreatic cancer: combine with gemcitabine for 100mg /d at least 1h before meals or 2h after meals; continue taking the drug until the disease get progression or the emergence of intolerable toxicity response when the patients should withdraw the drug.
Upon severe liver insufficiency, the drug dosage should be reduced or temporarily discontinued, elderly patients do not have to adjust the dose. | [Clinical evaluation]
Erlotinib is another tyrosine kinase inhibitor for the treatment of NSCLC after imatinib. The phase I clinical trial results have shown that its main toxicity and side effects are dose-dependent rash and diarrhea. Other rare side effects also include headache, nausea and vomiting.
Phase II trial takes erlotinib as a second line antineoplastic drugs with the efficacy being comparable as the second-line chemotherapy drug docetaxel. The Phase III randomized controlled trial (BR21) was performed in patients with NSCLC who had failed in the primary or secondary chemotherapy (locally advanced and distant metastases). Study group applied erlotinib in a dosage of 150mg daily for treatment of a total of 488 cases. A total of 243 cases were treated with placebo in the control group. The result: the median overall survival rate was 6.7 months in the treatment group and 4.7 months in the control group (P <0.001, HR = 0.73). The 1-year survival rate was 31.2% in the treatment group and 21.5% in the control group; the time of progression was 9.9 weeks in treatment group and 7.9 weeks in control group while the improvement of symptoms in patients with erlotinib was more obvious.
Based on the results of BR21 research, a number of phase Ⅲ clinical studies have been carried out successively. TRIBUTE clinical trial combined the Erlotinib with chemotherapy in an attempt to compare whether the efficacy of its combination with chemotherapy is better than chemotherapy alone. For the treatment group, chemotherapy (carboplatin plus paclitaxel) + erlotinib were combined; for the control group, the same chemotherapy was used alone. A total of 1059 patients with advanced NSCLC were included in the study. The efficacy in the research result: 21.5% for research group; 19.3% for the control group. The median survival time was 10.8 months in the study group and 10.6 months in the control group. The time to tumor progression (TTP) was 5.1 months in the study group and 5.0 months in the control group. Another TALENT study also focused on whether the combination of erlotinib and chemotherapy (gemcitabine + cisplatin) can improve the efficacy of chemotherapy and have included a total of 1172 cases of NSCLC patients. The results also did not show that erlotinib can significantly improve the efficacy of chemotherapy. | [Adverse reactions and precautions]
Common adverse reactions include rash, fever, anorexia, indigestion, nausea, vomiting, diarrhea, constipation and abdominal pain, fatigue, weight loss and edema, bone pain and muscle pain, dyspnea, elevated transaminases. In rare cases, it was observed of bone marrow suppression. Oral administration of warfarin may lead to unexpectedly increased international standardization ratio. There are occasionally chills, cough, stomatitis, keratoconjunctivitis, anxiety and neurological diseases, elevated bilirubin. In rare cases, there are microangiopathies hemolytic anemia and thrombocytopenia. Cough and fever may be associated with interstitial lung disease, if it occurs, the drugs should be discontinued. Cytochrome P450 enzymes affect the metabolism of the product. Its inhibitor ketoconazole or agonist rifampicin can change the plasma concentration of this product, resulting in increased toxicity or reduced toxicity. Patients of co-administration of warfarin should subject to closely monitoring of the international normalized ratio. | [What is molecular targeted therapy?]
Molecular targeted therapy is no longer a new term. Scientists are constantly exploring the pathogenesis of cancer molecular biology, realizing that if specific changes in cancer can be given by a powerful blow, it will greatly improve the treatment effect, triggering changes in anti-cancer treatment concept. In recent years, new molecular targeted drugs in clinical practice have achieved remarkable results. The practice has shown that the correctness and feasibility of the molecular targeted therapy theory, putting the cancer treatment to an unprecedented new stage.
Depending on the target and nature of the drug, the drugs of the major target molecule therapy can be divided into the following categories:
(1) Small molecule tyrosine kinase inhibitors of epidermal growth factor receptor (EGFR): such as gefitinib (Iressa), Erlotinib (Tarceva);
(2) anti-EGFR monoclonal antibody: such as cetuximab ();
(3) Anti-HER-2 monoclonal antibody: such as Herceptin (Trastuzumab);
(4) Bcr-Abl tyrosine kinase inhibitors: such as imatinib;
(5) Vascular endothelial growth factor receptor inhibitors: such as Bevacizumab (Avastin);
(6) anti-CD20 monoclonal antibody: such as rituximab (Rituximab);
(7) IGFR-1 kinase inhibitors such as NVP-AEW541;
(8) mTOR kinase inhibitors: such as CCI-779;
(9) Ubiquitin-proteasome inhibitors: such as Bortezomib;
(10) Other: such as Aurora kinase inhibitors, histone deacetylase (HDACs) inhibitors. |
|
|