| Identification | More | [Name]
Fosinopril | [CAS]
98048-97-6 | [Synonyms]
4-cyclohexyl-1-[2-[(2-methyl-1-propanoyloxy-propoxy)-(4-phenylbutyl)phosphoryl]acetyl]
FOSINOPRIL,(4S)-4-CYCLOHEXYL-1-[(RS)-2-METHYL-1-(PROPIONYLOXY)PROPOXY]-(4-PHENYLBUTYL)PHOSPHINYLACETYL-L-PROLINE
L-Proline, 4-cyclohexyl-1-(R)-(1S)-2-methyl-1-(1-oxopropoxy)propoxy(4-phenylbutyl)phosphinylacetyl-, (4S)-
fosenopril
[1[S^<*>^ (R^<*>^)]���,2α����,4β]-4-Cyclohexyl-1-[[[2-methyl-1-(1-oxopropoxy)-propoxy](4-phenylbutyl)phosphinyl]acetyl]-L-proline
Secorvas
SQ-28555
Staril
4β-Cyclohexyl-1-[[[(S)-2-methyl-1-(1-oxopropoxy)propoxy](4-phenylbutyl)phosphinyl]acetyl]-L-proline | [EINECS(EC#)]
1806241-263-5 | [Molecular Formula]
C30H46NO7P | [MDL Number]
MFCD00870759 | [Molecular Weight]
563.66 | [MOL File]
98048-97-6.mol |
| Questions And Answer | Back Directory | [Angiotensin converting enzyme inhibitor]
Angiotensin converting enzyme inhibitor (ACEI) is a therapeutic drug that has been widely attached importance to by scholars both at home and abroad in recent years to resist hypertension and kidney damage. The WHO/ISH1999 guidelines for the treatment of hypertension have included ACEI in the 6 major drugs for the treatment of hypertension. It inhibits vasodilatation by inhibiting renin angiotensin system, reducing adrenaline released from adrenal nerve terminals, reducing endothelin formation and inhibiting bradykinin decomposition and inactivation and angiectasis. It also promotes the synthesis of prostaglandins with vasodilating effect, reduces aldosterone secretion, decreases the retention of water and sodium, and increases renal blood flow. It has a significant protective effect on kidney. Fosinopril is the third generation of ACEI. After oral administration, it will be hydrolyzed into an active Fosinoprilat in the gastrointestinal mucosa and liver. The latter can inhibit angiotensin-converting enzyme, reduce the production of angiotensin II, therefore decreasing the peripheral resistance, decreasing aldosterone secretion and increasing plasma renin activity. Fosinopril also inhibits the degradation of bradykinin and reduces vascular resistance, thus reducing the effect of blood pressure.
This product expands arteries and veins at the same time. This not only reduces peripheral vascular resistance (afterload), but also reduces capillary wedge pressure (preload), thereby improving cardiac output and can be used for congestive heart failure. Oral absorption is rapid and complete and the absorption rate is about 36%, which is not affected by food. The time to reach peak is not related to the dose, usually in 3 hours. The half-life is about 12 hours, and 14 hours for the patients with heart failure. The plasma protein binding rate of Fosinoprilat is more than 95%, which can be secreted by milk. The single dose will take effect 1 hour after oral administration and the maximum effect can be achieved 2~4 hours after oral administration. The effect can be maintained for 24 hours. 44% ~ 50% of this product is removed from the kidneys, 46% to 50% can be excreted from the intestines after removal of the liver. In hemodialysis and peritoneal dialysis, the scavenging amount of this product is only 2% and 7% of urine clearance.
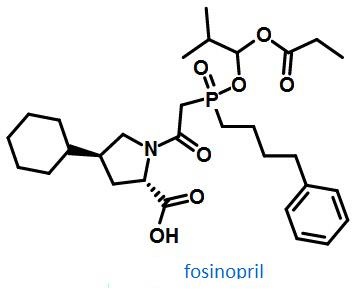
Figure1 The structural formula of fosinopril | [Precaution]
- Cautious use in the following cases: ①Autoimmune diseases (such as severe systemic lupus erythematosus):The chances of leukocyte or granulocytic reduction are increased at this time. ② Myelosuppression. ③ Cerebral or coronary artery insufficiency: ischemia can be aggravated by lowering blood pressure. ④ ]hyperkalemia. ⑤ Renal dysfunction: it will increase blood potassium, reduce leukocyte and granulocytic, and lead to the retention of the product.⑥ Liver dysfunction: reducing the metabolism of this product in the liver.⑦ Those with strict diet limiting sodium salt or dialysis treatment: The first dose of this product may result in sudden and severe hypotension.
- The administration of this product may result in the cases as follows: ①The concentration of blood urea nitrogen and creatinine is increased, which is often temporary. It is easy for this case to occur when there is a rapid decline in blood pressure in patients with kidney or severe hypertension.②There will be an occasional increase of serum liver enzyme. ③ A slight increase in blood potassium, especially in those with renal dysfunction.
- The follow-up examination during the administration of this product: ①For patients with renal dysfunction or leukocyte deficiency, the white blood cell count and classification count are checked every 2 weeks in the first 3 months, and then checked regularly thereafter. ② Urine protein examination, once a month.
- The antihypertensive effect of this product is the same in the standing position and the supine position.
- For those who have used diuretics, they should stop using diuretics for 2~3 days before using this product. But those with severe or malignant hypertension are exceptional. They should use this product at a low dose and carefully increase the dose under close observation.
- Medication should be stopped during the period of vascular edema, and adrenaline is injected subcutaneously and hydrocortisone is injected intravenously.
- FDA is classified as grade C for the safety of pregnancy.
If in the middle and late pregnancy, the drug is D grade.
| [Application]
- Light, medium and severe hypertension: It can be prioritized as the first drug, and can also be used when other antihypertensive drugs have poor curative effect.
- Myocardial ischemia: it can reduce the oxygen consumption of myocardium, increase the oxygen supply of myocardium, double the treatment of myocardial ischemia, reduce the frequency of angina pectoris and the dosage of nitrate.
- Treatment of heart failure: it can be used as the first choice by dilating blood vessels and reducing sodium retention.
| [Prohibited use]
- It is forbidden for those who are allergic to this product or other angiotensin converting enzyme inhibitors.
- Those with solitary kidney, transplant kidney, bilateral renal artery stenosis and renal dysfunction are forbidden to use this product.
| [Interaction]
This product can reduce the decrease of potassium induced by thiazide diuretics. Its combination with potassium diuretic or potassium supplement can increase the risk of hyperkalemia. If this kind of drug should be used at the same time, the patient's serum potassium needs to be monitored regularly. Acid resistant drugs may affect the absorption of this product. If taken, it must be separated at least two hours.Non steroidal anti-inflammatory drugs can affect the effect of this product on lowering blood pressure. But at the same time, the use of this product and steroidal anti-inflammatory drugs (including aspirin) do not increase the other clinical adverse reactions. Treatment with lithium can increase the concentration of serum lithium. The combined use of beta blockers, Methyldopa, calcium antagonists and diuretics can increase the efficacy of antihypertensive drugs.
|
| Hazard Information | Back Directory | [Originator]
Fosinopril Sodium,Bristol-Myers Squibb | [Uses]
mydriatic, | [Definition]
ChEBI: A phosphinate ester-containing N-acyl derivative of (4S)-cyclohexyl-L-proline. It is used for the treatment of hypertension and heart failure. A pro-drug, it is hydrolysed in vivo to the corresponding phosphin
nc acid, fosinoprilat, which is the active metabolite. | [Manufacturing Process]
To a solution of 4-phenylbutyl phosphinic acid (2.0 g, 0.01 mole) in
chloroform (40 ml) was added triethylamine (3.2 ml, 0.022 mole) and the
mixture was cooled in an ice bath to 0°C. Trimethylsilyl chloride (2.8 ml,
0.022 mole) was added to the above solution dropwise, followed by benzyl
bromoacetate (1.6 ml, 0.011 mole). The ice bath was removed and the
mixture stirred at room temperature for 5 hours and poured into 10%
aqueous HCl (30 ml) and crushed ice (20 g). After shaking the mixture in a
separatory funnel, the chloroform layer was separated and the aqueous layer
was extracted with dichloromethane. The combined organic phase was washed
with brine, dried over anhydrous sodium sulfate and the solvents removed in
vacuum. The resulting crude thick oil (3.5 g) was dissolved in 30 ml ether,
hexane was added dropwise to get a turbid solution and the mixture was left
at room temperature overnight to complete the crystallization. It was cooled
in the freezer for 2 hours, filtered and the solid was washed very thoroughly
with hexane (50 ml), ether (50 ml) and again hexane (50 ml), ether (50 ml)
in that order. The solid was vacuum dried to get 2.48 g (71%) of [hydroxy-(4-phenylbutyl)-phosphinyl]acetic acid, phenylmethyl ester, m.p. 68-70°C. TLC
(Silica gel, CH2Cl2:MeOH:HOAc (20:1:1)) shows a single spot.
A solution of 50 g (0.14 mole) of [hydroxy-(4-phenylbutyl)-phosphinyl]acetic
acid, phenylmethyl ester in 300 ml of dry CHCl3 was treated with 28.6 g (0.28
mole) of Et3N, 35.6 g (0.21 mole) of 1-chloroisobutyl propionate, 12.0 g
(0.035 mole) of (n-Bu)4NHSO4 and 5.3 g (0.035 mole) of NaI. The above
mixture was stirred and heated to mild reflux for 20 hours, then cooled and
the solvent evaporated in vacuo. The oil residue was dissolved in 150 ml of
ether and washed with 150 ml of water. The aqueous wash was extracted with
150 ml of ether. The combined ether solutions were washed with 5% NaHCO3,
10% NaHSO3 and brine. After drying (MgSO4) the ether was evaporated in
vacuo to give 57.0 g (83%) of crude [[2-methyl-1-(1-oxopropoxy)propoxy](4-
phenylbutyl)phosphinyl]acetic acid, phenyl methyl ester as an oil product.
A solution of 57.0 g (0.12 mole) of [[2-methyl-1-(1-oxopropoxy)propoxy](4-
phenylbutyl)phosphinyl]acetic acid, phenyl methyl ester in 300 ml of ethyl
acetate was treated with 3.0 g of 10% Pd/C and hydrogenated on the Parr
apparatus (45 psi) for 4 hours. The mixture was filtered through Hyflo and the
solution was extracted with 5% NaHCO3. The aqueous extracts were washed
with ether, cooled to 5°C and treated with 36 ml of HOAc. The product was
extracted into ethyl acetate, dried (MgSO4) and the solvent was evaporated in
vacuo. The residue was dissolved in 300 ml of toluene and the solvent was
evaporated in vacuo to remove last traces of acetic acid. The oil residue
became semi-solid on standing at room temperature. The yield of the product
of debenzoylation - 2-[carboxymethyl)-(4-phenylbutyl)-phosphinoyloxy]-2-
methylpropionic acid ethyl ester (racemic mixtures) was 39.8 g (72%).
A suspension of 10.0 g (0.026 mole) of [[2-methyl-1-(1-oxopropoxy)propoxy]
(4-phenylbutyl)phosphinyl]acetic acid in 50 ml of isopropyl ether was stirred
vigorously for 15 min, then kept at 5°C for 20 hours. The colorless product
was filtered, washed with a small amount of cold isopropyl ether to give 5.0 g
of [[2-methyl-1-(1-oxopropoxy)propoxy](4-phenylbutyl)phosphinyl]acetic acid
(A/B isomer, racemic mixture) , m.p. 87-89°C. The filtrate was evaporated in
vacuo and retained for isolation of isomer C/D. A solution of the above
material in 110 ml of hot isopropyl ether was filtered through a hot glass
funnel (glass wool). The cooled solution gave 4.6 g (92%) of desired product,
m.p. 90-92°C.
To a vigorously stirred suspension of 980 g (3.33 mol) of l-cinchonidine in 6 L
of ethyl acetate maintained at 45°C was gradually added 1275.5 g (3.33 mol)
of A/B isomer mixture and stirring then continued for an additional 2.5 hours
while the resulting suspension of salt was gradually heated to 70°C when
complete solution was obtained. After filtration (Hyflo) from a small amount of
insoluble material, the solution was seeded and cooled. The crystalline product
which separated was then filtered, washed with 1200 ml of 1:1 ethyl
acetate/isopropyl ether, and dried in vacuo to give 1897.2 g of cinchonidine
salt enriched in the B-isomer, m.p. 106-109°C, [α]D = -59.3° (c = 1,
methanol). This material was combined with 136.8 g of similarly prepared
material (from 0.412 mol of A/B isomer) and the total quantity (2014 g)
recrystallized from 10.18 L of boiling ethyl acetate to afford after filtration,
washing with 1500 ml of the same solvent mixture used before, and drying in
vacuo 1162 g (92%) of [[2-methyl-1-(1-oxopropoxy)propoxy](4-phenylbutyl) phosphinyl]acetic acid (Resolution; isomer B), cinchonidine salt (1:1), m.p.
120-122°C (dec.), [α]D= -45° (c = 1, methanol), [α]365 = -185.5° (c = 1,
methanol). A sample (10 g) was recrystallized twice from acetonitirle and
three times from ethyl acetate additionally to give salt of m.p. 125-126°C
(dec.), [α]D= -42.2°.
A slurry of methyl-1-(1-oxopropoxy)propoxy](4-phenylbutyl)phosphinyl]acetic
acid (B-isomer), dried in vacuo at room temperature for 72 hours, (230.4 g,
0.6 moles) and hydroxybenzotriazole hydrate, dried, in vacuo at 80°C for 24
hours, (101.1 g, 0.66 mole) dichloromethane (sieved dried) (6 L) was chilled
in an ice/acetone bath and treated with N,N-dicyclohexylcarbodiimide (136 g,
0.66 mole). The mixture was warmed to room temperature and stirred for 3
hours. The mixture was then chilled in ice/acetone and treated with (trans)-4-
cyclohexyl-L-proline, hydrochloride (154.2 g, 0.66 mole) followed by
diisopropylethylamine (170.7 g, 1.32 mole). The reaction mixture was stirred
at room temperature for 18 hours. The mixture was then chilled, treated with
water (1 L) and concentrated in vacuo to remove dichloromethane. The
residue was diluted with ether (3600 ml) and water (3600 ml) and filtered.
The filtrate was brought to pH = 1.8 with 10% hydrochloric acid. The ether
layer was separated and the aqueous layer washed with ethyl acetate (3 x 2
L). The combined organic layers were washed with 5% KHSO4 (3 x 1 L), water
(3 x 1 L) and brine (1 L), dried over magnesium sulfate and concentrated in
vacuo to yield 398.9 g of crude [R,1S,4S]-4-Cyclohexyl-1-[[[2-methyl-1-(1-
oxopropoxy)propoxy](4-phenylbutyl)phosphinyl]acetyl]-L-proline, monosodium
salt (isomer B). The crude product was dissolved in acetone (4393 ml),
treated with a solution of 2-ethyl hexanoic acid, sodium salt (117.3 g) in
acetone (1468 ml), then stirred at room temperature overnight. The resultant
precipitate was collected by filtration, washed with acetone (3 x 400 ml) and
hexane (1 L) then dried in vacuo. Yield 277 g, m.p. 195-196°C, [α]D= -5.1°
(MeOH, c = 2), HI = 99.8%. Isomer "A" was not detectable. | [Brand name]
Monopril (Bristol-Myers Squibb). | [Therapeutic Function]
Antihypertensive |
|
| Company Name: |
LGM Pharma
|
| Tel: |
1-(800)-881-8210 |
| Website: |
www.lgmpharma.com |
| Company Name: |
China Langchem Inc.
|
| Tel: |
0086-21-58956006 |
| Website: |
www.approvedhomemanagement.com/ShowSupplierProductsList19141/0.htm |
|