| | Bromazepam Basic information |
| Product Name: | Bromazepam | | Synonyms: | 7-BROMO-5-[2-PYRIDYL]-3H-1,4-BENZODIAXEPIN-2[1H]-ONE;BROMAZEPAM;1,3-Dihydro-7-bromo-5-(2-pyridyl)-2H-1,4-benzodiazepin-2-one;1,3-dihydro-7-bromo-5-(2-pyridyl)-2h-4-benzodiazepin-2-one;2H-1,4-Benzodiazepin-2-one, 1,3-dihydro-7-bromo-5-(2-pyridyl)-;2H-1,4-Benzodiazepin-2-one, 7-bromo-1,3-dihydro-5-(2-pyridinyl)-;2H-1,4-Benzodiazepin-2-one, 7-bromo-1,3-dihydro-5-(2-pyridyl)-;7-Bromo-1,3-dihydro-5-(2-pyridyl)-2H-1,4-benzdiazepin-2-one | | CAS: | 1812-30-2 | | MF: | C14H10BrN3O | | MW: | 316.15 | | EINECS: | 217-322-4 | | Product Categories: | Heterocycles;Intermediates & Fine Chemicals;Pharmaceuticals | | Mol File: | 1812-30-2.mol | 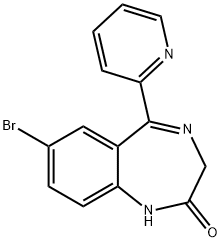 |
| | Bromazepam Chemical Properties |
| Melting point | 237-238.5oC (dec.) | | Boiling point | 490.7±45.0 °C(Predicted) | | density | 1.5573 (rough estimate) | | refractive index | 1.6520 (estimate) | | Fp | 11 °C | | storage temp. | 2-8°C | | solubility | Practically insoluble in water, slightly soluble or sparingly soluble in ethanol (96 per cent) and in methylene chloride. | | form | A crystalline solid | | pka | pKa 2.5(spectro,
polarographic) (Uncertain);5.2(spectro,
polarographic) (Uncertain);11.8(spectro,
polarographic) (Uncertain) | | CAS DataBase Reference | 1812-30-2(CAS DataBase Reference) | | NIST Chemistry Reference | Bromazepam(1812-30-2) |
| | Bromazepam Usage And Synthesis |
| Description | Bromazepam is a psychoactive benzodiazepine derivative and prescription drug that is commonly abused and often used in ‘drug-facilitated crimes’. This product is intended for forensic and research applications. | | Chemical Properties | Crystalline Solid | | Originator | Lexotan,Roche,Italy,1975 | | Uses | Controlled substance (depressant). Anxiolytic. | | Definition | ChEBI: Bromazepam is an organic molecular entity. | | Manufacturing Process | Example: 32.8 grams of 2-(2-aminobenzoyl)-pyridine and 200 cc of acetic
anhydride were stirred at room temperature for 3 hours and then permitted to
stand overnight. Evaporation to dryness and digestion of the residue with 200
cc of water containing a little sodium bicarbonate to make the pH slightly
alkaline gave 2-(2-acetamidobenzoyl)-pyridine as a light tan powder, which
upon crystallization from methanol formed colorless crystals melting at 151°-
153°C.
A solution of 8.6 cc of bromine in 100 cc of acetic acid was added slowly over
a 3.5 hour period to a stirred solution of 38.5 grams of 2-(2-
acetamidobenzoyl)-pyridinein 250 cc of acetic acid. The dark solution was
stirred for another 3 hours, permitted to stand over night, stirred for 1 hour
with N2 sweeping, and evaporated at diminished pressure in the hood. The
gummy residue (75 grams) was treated with water and ether, made alkaline
with dilute sodium bicarbonate solution, and separated. Both phases contained
undissolved product which was filtered off. Additional crops were obtained by
further extraction of the aqueous phase with ether and evaporation of the
resulting ether solutions. All these materials were recrystallized from methanol
(decolorizing carbon added) yielding 2-(2-acetamido-5-bromobenzoyl)-
pyridineas yellow crystals melting at 131.5°-133°C.
20.85 grams of 2-(2-acetamido-5-bromobenzoyl)-pyridinein 250 cc of 20%
hydrochloric acid in ethanol were heated to reflux for 2 hours. 100 cc of
alcohol were added after one hour to maintain fluidity. The mixture stood
overnight, was chilled and filtered to give 20.5 grams of colorless crystalline
2-(2-amino-5-bromobenzoyl)-pyridinehydrochloride. Digestion of this hydrochloride with 0.5liter hot water hydrolyzed this product to the free base,
2-(2-amino-5-bromobenzoyl)-pyridine which formed yellow crystals, melting
at 98°-100°C. Evaporation of the alcoholic mother liquor, water digestion of
the residue, and alkalization of the water digests afforded additional crops of
2-(2-amino-5-bromobenzoyl)pyridine.
0.145 kg of 2-(2-amino-5-bromobenzoyl)-pyridine, was dissolved in 2.0 liters
of glacial acetic acid. The resultant solution was placed in a 3 liter, 3-necked,
round bottom flask fitted with a stirrer, thermometer and dropping funnel. The
system was protected by a drying tube filled with anhydrous calcium chloride.
To the solution, with stirring at room temperature, were carefully added 46.7
ml of bromoacetyl bromide. After the addition was completed, the stirring was
continued for two hours. The mixture was then warmed to 40°C, stirred at
that temperature for 1.5 hours, chilled and filtered. The residue, after being
washed with glacial acetic acid, was dried in vacuo over flake potassium
hydroxide to give 2-(2-bromoacetamido-5-bromobenzoyl)-
pyridinehydrobromide orange crystals, MP 205°-206°C, dec.
The hydrobromide was hydrolyzed to the free base as follows: 0.119 kg of 2-
(2-bromoacetamido-5-bromobenzoyl)-pyridine hydrobromide was stirred with
1.2 liters of cold water for 3.5 hours. The mixture was chilled and filtered, and
the residue washed with cold water and dried to give 2-(2-bromoacetamido-5-
bromobenzoyl)-pyridine, MP 101°C (sinters), 103°-106°C, dec.
93.0 grams of 2-(2-bromoacetamido-5-bromobenzoyl)-pyridinewas carefully
added to 0.5 liter of anhydrous ammonia in a 1 liter, 3-necked, round bottom
flask equipped with stirrer and reflux condenser and cooled by a Dry Iceacetone
bath. The system was protected from moisture by a drying tube
containing anhydrous calcium chloride. After stirring for 2 hours, the cooling
bath was removed. The mixture was then stirred for 6 hours, during which
time the ammonia gradually boiled off. 0.4 liter of water was added to the
solid residue and stirrind was resumed for about 2 hours. The solid was then
filtered off, washed with water and dried in vacuo over potassium hydroxide
flakes. The residue was dissolved on a steam bath in 1.4 liters of ethyl
alcohol-acetonitrile (1:1) (decolorizing charcoal added). The solution was
filtered hot and the filtrate chilled overnight. The crystalline deposit was
filtered off, washed with cold ethyl alcohol and dried in vacuo over flake
potassium hydroxide to give 54.2 grams. 7-Bromo-1,3-dihydro-5-(2-pyridyl)-
2H-1,4-benzodiazepin-2-one, MP 238°C (sinters), 239°-240.5°, dec. Further
processing of the mother liquor yielded additional product. | | Brand name | Lectopam (Roche,
Puerto Rico). | | Therapeutic Function | Tranquilizer | | Enzyme inhibitor | This lipophilic valium-like benzodiazepine (FW = 316.20 g/mol; CAS 1812- 30-2; IUPAC Name: 7-bromo-5-(pyridin-2-yl)-1H-benzo[e][1,4]diazepin- 2(3H)-one), is a long-lasting anxiolytic agent used to treat anxiety or panic states. Like diazepam, bromazepam is a positive allosteric modulator of GABAA receptors, promoting GABA binding, which in turn increases the total conduction of chloride ions across the neuronal cell membrane. Like other benzodiazepines, bromazepam also inhibits the acetylcholine receptoroperated potassium current. Bromazepam is mainly metabolized by oxidative pathways within the liver. |
| | Bromazepam Preparation Products And Raw materials |
|