| Structure |
Chemical Name |
CAS |
MF |
 |
Gatifloxacin USP IMpurity C |
|
|
 |
Enrofloxacin hydrochloride |
|
C19H23ClFN3O3 |
 |
Hydrochloric acid pazufloxacin |
|
|
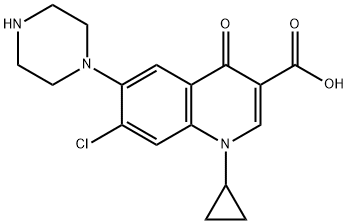 |
Ciprofloxacin EP IMpurity D |
133210-96-5 |
C17H18ClN3O3 |
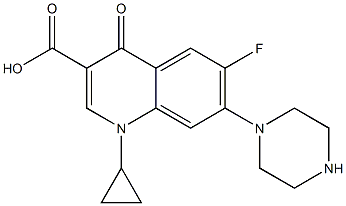 |
Ciprofloxacin IMpurity E |
|
C17H18FN3O3 |
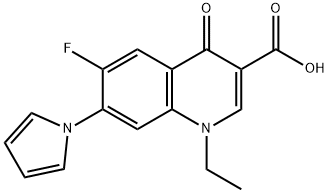 |
Irloxacin |
91524-15-1 |
C16H13FN2O3 |
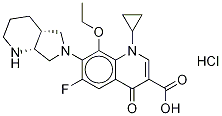 |
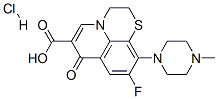
8-Ethoxy Moxifloxacin Hydrochloride |
|
C22H27ClFN3O4 |
 |
Rufloxacin hydrochloride |
106017-08-7 |
C17H19ClFN3O3S |
 |
Difloxacin mesylate |
|
|
 |
Moxifloxacin for peak identification |
|
|
 |
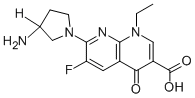
Norfloxacin Lactate Injection |
|
|
 |
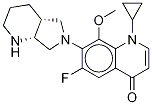
Esafloxacin |
79286-77-4 |
C15H17FN4O3 |
 |
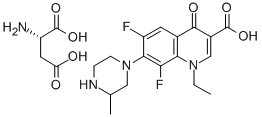
Decarboxy Moxifloxacin |
|
C20H24FN3O2 |
 |
LOMEFLOXACIN, ASPARTATE |
211690-33-4 |
C17H19F2N3O3.C4H7NO4 |
 |
SparfloxacinLactate |
|
C19H22F2N4O3·C3H6O3 |
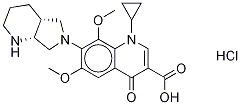 |
6,8-Dimethoxy Moxifloxacin Hydrochloride |
|
C22H28ClN3O5 |
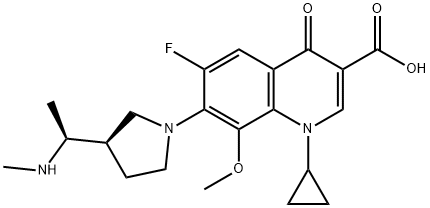 |
permafloxacin |
143383-65-7 |
C21H26FN3O4 |
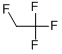 |
1,2,2,2-tetrafluoroethane |
29759-38-4 |
C16H18FN3O3C6H5NO3 |
 |
Enoxacin gluconate |
|
|
 |
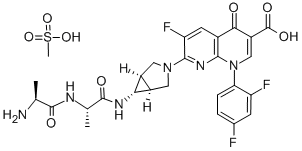
IMp. A (EP): 7-Chloro-1-ethyl-6-fluoro-4-oxo-1,4-dihydroquinoline-3-carboxylic Acid |
|
|
 |
Alatrofloxacin |
146961-76-4 |
C26H25F3N6O5 |
 |
Norfloxacin glutamic acid |
|
|
 |
Intermediate of Lomefloxacin |
|
|
 |
PEFLOXACIN MESILATE DIHYDRATE |
|
|
 |
ENROFLOXACIN BASE |
|
|
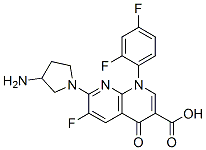 |
Tosufloxacin |
108138-46-1 |
C19H15F3N4O3 |
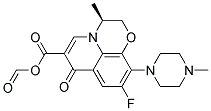 |
Levofloxacin Methylate |
|
C19H20FN3O5 |
 |
NORFLOXACIN BASE |
|
|
 |
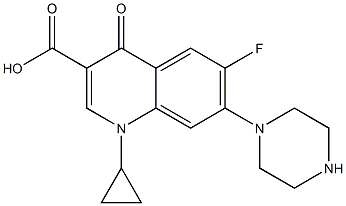
Alatrofloxacin |
|
|
 |
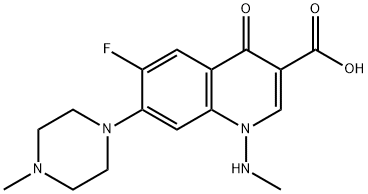
Ciprofloxacin IMpurity A |
|
C17H18FN3O3 |
 |
amifloxacin |
86393-37-5 |
C16H19FN4O3 |
 |
Gatifloxacin mesylate |
|
|
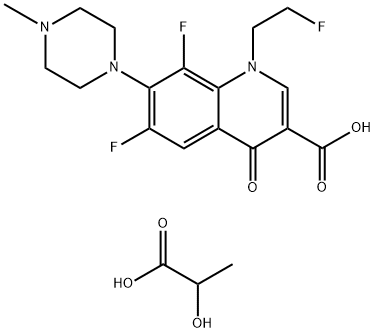 |
FLEROXACINLACTATE |
896139-71-2 |
C20H24F3N3O6 |
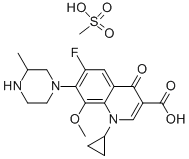 |
GATIFLOXACIN MESYLATE |
316819-28-0 |
C19H22FN3O4.CH4O3S |
 |
Enrofloxacin nicotinate |
|
|
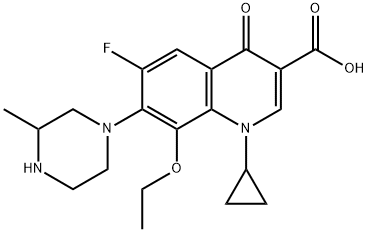 |
3-Quinolinecarboxylic acid, 1-cyclopropyl-8-ethoxy-6-fluoro-1,4-dihydro-7-(3-Methyl-1-piperazinyl)-4-oxo- |
182868-72-0 |
C20H24FN3O4 |
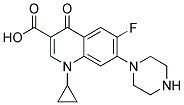 |
CIPROFLOXACIN IMPURITY A |
|
C17H18FN3O3 |
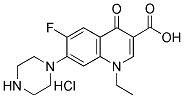 |
NORFLOXACIN HYDROCHLORIDE |
|
C16H19ClFN3O3 |