Telmisartan Chemische Eigenschaften,Einsatz,Produktion Methoden
R-S?tze Betriebsanweisung:
R36/37/38:Reizt die Augen, die Atmungsorgane und die Haut.
S-S?tze Betriebsanweisung:
S22:Staub nicht einatmen.
S24/25:Berührung mit den Augen und der Haut vermeiden.
S36:DE: Bei der Arbeit geeignete Schutzkleidung tragen.
S26:Bei Berührung mit den Augen sofort gründlich mit Wasser abspülen und Arzt konsultieren.
Beschreibung
Telmisartan is an angiotensin receptor blocker (ARB), It is used in the treatment of hypertension also effective in cardiovascular risk reduction.Telmisartan blocks the action of angiotensin II (Ang II), the primary effector molecule of the renin-angiotensin-aldosterone system (RAAS). It is the sixth of this class of 《sartans》 to be marketed after the lead compound Losartan. Its long lasting effect (24h half-life) could be the main difference with other angiotensin II antagonists. Unlike several other agents in this category, its activity does not depend upon transformation into an active metabolite, the 1-O-acylglucuronide being the principal metabolite found in humans. Telmisartan is a potent competitive antagonist of AT1 receptors that mediate most of the important effects of angiotensin II while lacking affinity for the AT2 subtypes or other receptors involved in cardiovascular regulation.
Chemische Eigenschaften
White or off white crystalline powder
Verwenden
Telmisartan, an angiotensin II receptor antagonist, is an effective medication for the treatment of hypertension. It can be used alone or in combination with other antihypertensive drugs. Additionally, it is beneficial in the treatment of diabetic nephropathy in hypertensive individuals with type 2 diabetes mellitus. Telmisartan is also used to address congestive heart issues.
Definition
ChEBI: Telmisartan is a member of the class of benzimidazoles used widely in the treatment of hypertension. It has a role as an antihypertensive agent, an angiotensin receptor antagonist, an EC 3.4.15.1 (peptidyl-dipeptidase A) inhibitor, a xenobiotic and an environmental contaminant. It is a member of biphenyls, a member of benzimidazoles and a carboxybiphenyl.
Allgemeine Beschreibung
Telmisartan, 4'-[(1,4'-dimethyl-2'-propyl[2,6'-bi-1H-benzimidazol]-1'-yl)methyl]-[1,1'-biphenyl]-2-carboxylic acid (Micardis), does not appear to bear any structuralrelationship to this class, but there is actually a great dealof overlap in the chemical architecture with other agents. Thefirst, and most significant, difference is the replacement of theacidic tetrazole system with a simple carboxylic acid. Thisacid, like the tetrazole, plays a role in receptor binding. Thesecond difference is the lack of a carboxylic acid near the imidazolenitrogen that also contributes to receptor binding.As with irbesartan, however, there is not a need for this groupto be acidic but, rather, to be one that participates in receptorbinding. The second imidazole ring, much like a purine basein deoxyribonucleic acid (DNA), can hydrogen bond with theangiotensin II receptor.
Synthese
Telmisartan can be prepared in eight steps starting with methyl 4-amino-3-methyl benzoate; the first and second cyclization into a benzimidazole ring occur at steps 4 and 6 respectively.
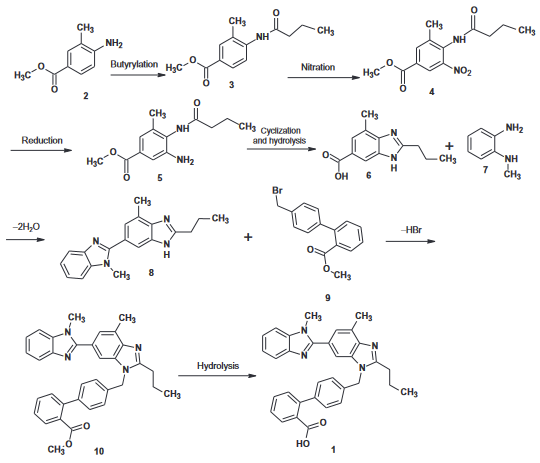
Telmisartan Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Downstream Produkte

