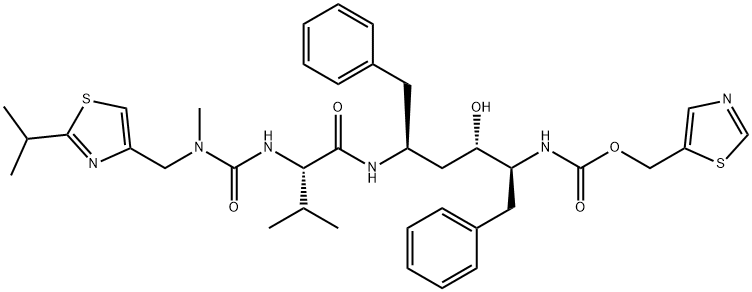
Ritonavir synthesis
- Product Name:Ritonavir
- CAS Number:155213-67-5
- Molecular formula:C37H48N6O5S2
- Molecular Weight:720.94
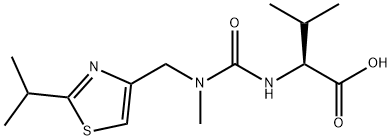
154212-61-0
221 suppliers
$5.00/1g
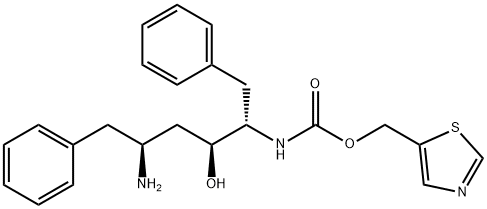
144164-11-4
117 suppliers
$96.00/1g

155213-67-5
589 suppliers
$5.00/10mg
Yield:155213-67-5 91.5%
Reaction Conditions:
Stage #1: N-((N-Methyl-N-((2-isopropyl-4-thiazolyl)methyl)amino)-carbonyl)-L-valinewith N-ethyl-N,N-diisopropylamine;diisopropyl-carbodiimide at 27; for 0.5 h;
Stage #2: (2S,3S,5S)-5-Amino-2-(N-((5-thiazolyl)-methoxycarbonyl)amino)-3-hydroxy-1,6-diphenylhexane for 7 h;Temperature;
Steps:
2 Preparation of Embodiment 2: ritonavir
In the 27 °C lower, the compound N - [N - methyl - N - [(2 - isopropyl -4 - thiazolyl) methyl] aminocarbonyl] - L - valine 37.6g (120.0mmol), 15.1g (120.0mmol) DIC (N, N' - diisopropyl carbodiimide), 35.0gN, N - diisopropyl ethylamine and 200 ml of cyclopentanone mixing 0.5h. The mixture into the to 42.6g (100mmol) (2S, 3S, 5S) -5 - amino -2 - (N - ((5 - thiazolyl) - methoxycarbonyl) amino) - 1, 6 - diphenyl -3 - hydroxy hexane 150 ml of cyclopentanone in stirring 7h, TLC detection, said technological, turn the reaction liquid 10% sodium chloride and purified water washing 350 ml × 2, abandoned to the aqueous layer, the organic phase adding anhydrous sodium sulfate drying 6h after-filtration, then the filtrate is heated to 45 - 50 °C, at the same time adding 0.8g activated carbon to decolorize the, stirring 15min, cooling to 20 - 30 °C filter, vacuum concentrated organic phase to be oily lito that wei Cupin, up into the oil objects in 230 ml butyl acetate, then heating to 60 °C, to dissolve it completely, natural cooling to 20 - 30 °C, and stirring 12h, filtering, cold-butyl acetate 40 ml washing the filter cake, vacuum 50 °C drying 6h, obtaining white crystalline grain product ritonavir 66.3g, yield 91.5%,
References:
CN106749085,2017,A Location in patent:Paragraph 0020; 0021; 0022; 0023
![2,5-DIOXOPYRROLIDIN-1-YL N-{N-[(2-ISOPROPYL-1,3-THIAZOL-4-YL)METHYL]-N-METHYLCARBAMOYL}-L-VALINATE](/CAS/GIF/224631-15-6.gif)
224631-15-6
1 suppliers
inquiry

144164-11-4
117 suppliers
$96.00/1g

155213-67-5
589 suppliers
$5.00/10mg
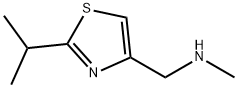
154212-60-9
140 suppliers
$45.00/50mg

155213-67-5
589 suppliers
$5.00/10mg
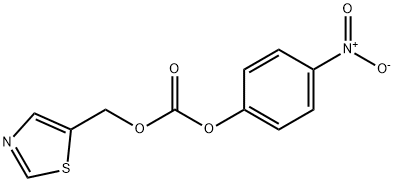
144163-97-3
235 suppliers
$9.00/5g

155213-67-5
589 suppliers
$5.00/10mg
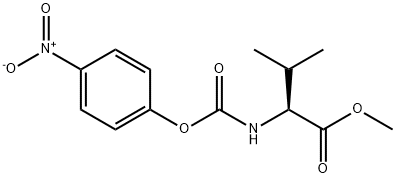
162537-10-2
17 suppliers
inquiry

155213-67-5
589 suppliers
$5.00/10mg