Versatile Acylation of N-Nucleophiles Using a New Polymer-Supported 1-Hydroxybenzotriazole Derivative
Abstract
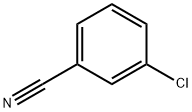
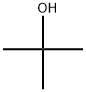
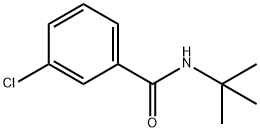
The synthesis of a new polymer-supported coupling reagent derived from 1-hydroxybenzotriazole is described. An aminomethylated polystyrene was functionalized by reaction with 3-nitro-4-chlorobenzenesulfonyl chloride (2) followed by treatement with hydrazine hydrate, to give the polymeric N-benzyl-1-hydroxybenzotriazole-6-sulfonamide (4).The polymeric reagent 4 was shown to be highly efficient for the synthesis of amides. The efficiency of 4 could be attributed to its high acidity, conferred by the sulfonyl moiety. The procedure for amide synthesis involves the formation of an activated ester on the derivatized polymer followed, in a second step, by treatment with an amine to generate the amide in solution. Simple filtration allows the separation of the product from the polymeric reagent which in this case plays the role of leaving group. An optimization study of this two-step procedure was performed. As amides are obtained in solution free of reaction byproducts, this method can be used in an automated procedure to recover them directly into a 96 well plate, ready to be used in high throughput screening assays. Thus 4 was shown to be particularly suitable for the high throughput parallel synthesis of amides libraries.