| 39% |
Stage #1: With Geobacillus stearothermophilus Tc-62 cyclomaltodextrin glucanotransferase In water at 55℃; for 50 h; Enzymatic reaction
Stage #2: With glucoamylase GLUCZYM AF6 In water for 24 h; Enzymatic reaction |
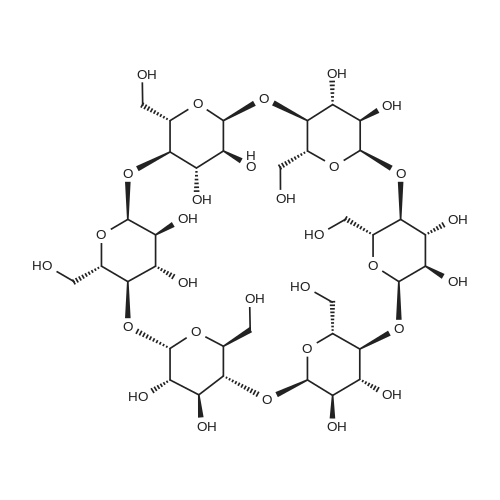
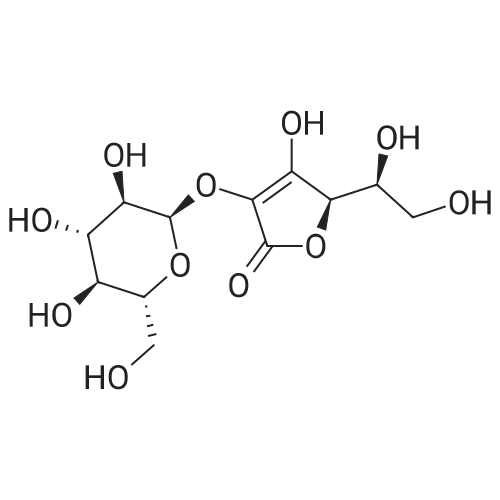
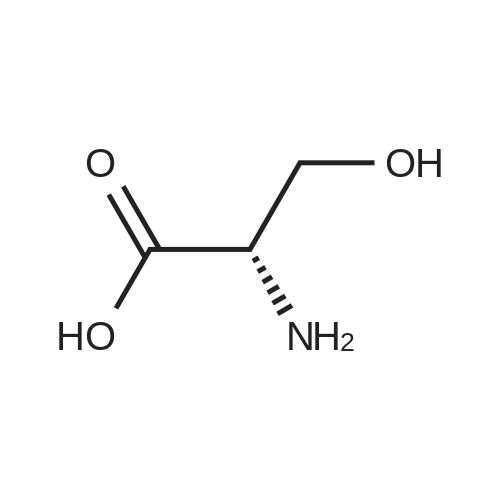
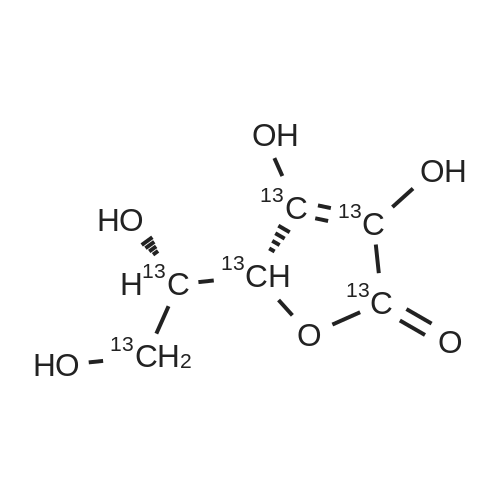
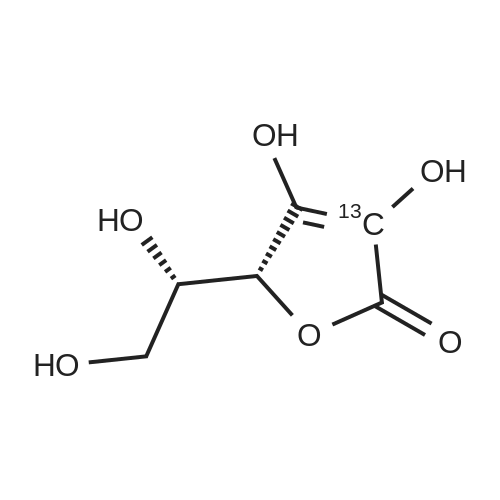
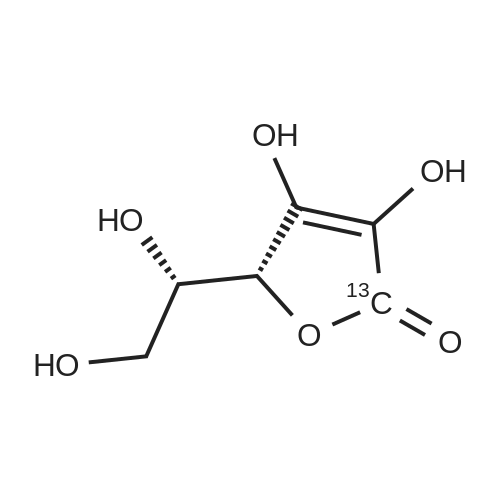
Test samples 10 to 15, having mutually different purities of ascorbic acid 2-glucoside as shown in Table 3, were prepared from an aqueous solution containing L-ascorbic acid and dextrin. Four parts by weight of "PINEDEX 100", a product name of a dextrin commercialized by Matsutani Chemical Industries Co., Ltd., Hyogo, Japan, was dissolved in 15 parts by weight of water by heating. Then, three parts by weight of L-ascorbic acid was admixed with the solution. Successively, the solution was admixed with 100 units/g dextrin, d.s.b., of a CGTase derived from Geobacillus stearothermophilus Tc-62 strain and 250 units/g dextrin, d.s.b., of an isoamylase specimen, commercialized by Hayashibara Biochemical Laboratories, Inc., Okayama, Japan, and subjected to an enzymatic reaction while keeping the solution at a pH of 5.5 and a temperature of 55°C for 50 hours to form ascorbic acid 2-glucoside. In addition, it can be speculated that α-glycosyl-L-ascorbic acids such as 2-O-α-maltosyl-L-ascorbic acid, 2-O-a-maltotriosyl-L-ascorbic acid, 2-O-α-maltotetraosyl-L-ascorbic acid, etc., would have been naturally formed in the reaction solution. After inactivating the remaining enzymes by heating, the reaction solution was adjusted to pH 4.5, admixed with 50 units/g dextrin, d.s.b., of "GLUCZYM AF6", a product name of a glucoamylase specimen commercialized by Amano Enzymes Inc., Aichi, Japan, and subjected to an enzymatic reaction for 24 hours for hydrolyzing the above α-glycosyl-L-ascorbic acids up to ascorbic acid 2-glucoside and hydrolyzing the remaining concomitant oligosaccharides up to D-glucose. At this stage, the reaction solution contained ascorbic acid 2-glucoside in a production yield of 39percent. The reaction solution was heated to inactivate glucoamylase, decolored with an activated charcoal, filtered, subjected to a column of a cation-exchange resin (H+-form) for desalting, and then subjected to an anion-exchange resin (OH--form) to absorb L-ascorbic acid and ascorbic acid 2-glucoside, followed by washing the resin with water to remove D-glucose and feeding 0.5 N hydrochloric acid solution to effect elution. The eluate was concentrated to give a solid content of about 50percent and then subjected to a column chromatography using "DOWEX 50WX4" (Ca2+-form), a product name of a strong-acid cation exchange resin commercialized by Dow Chemical Company. The concentrate was loaded on the column in a volume of about 1/50-fold of the wet resin volume, followed by feeding to the column refined water in a volume of 50-folds of the load volume of the concentrate at a linear velocity of 1 m/hour and fractionating the resulting eluate by 0.05-volume aliquots of the column volume. Thereafter, the composition of each fraction was measured on HPLC described in Experiment 1-2, and six fractions with an ascorbic acid 2-glucoside content of at least 80percent, d.s.b., were concentrated in vacuo to give respective solid concentrations of about 76percent. The resulting concentrates were respectively placed in a crystallizer, admixed with Test sample 1 in Experiment 1-1, as a seed crystal, in a content of two percent of each of the solid contents, d.s.b., followed by unforcedly cooling each concentrate from 40°C to 15°C over about two days while stirring gently to precipitate anhydrous crystalline ascorbic acid 2-glucoside. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 120K+ Compounds
120K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping