| 81% |
Product distribution / selectivity; |
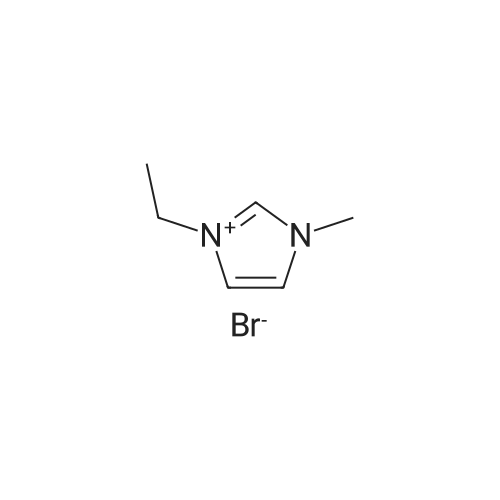
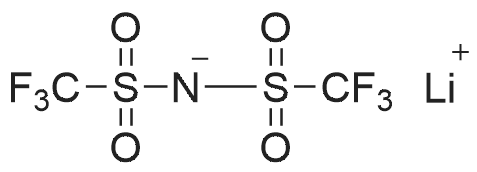
To 48.75 g (0.097 mol) of tris(diethylamino) n-butylaminophosphonium iodide obtained in B(h), an aqueous solution dissolving 28.7 g (0.100 mol) of LiTFSI in 200 ml of ultrapure water was added, and then the resultant mixture was stirred at 50C for 3 days. The resulting salt was extracted with 100 ml of CH2Cl2, and the water layer was further extracted with 50 ml of CH2Cl2. After five times of washing with ultrapure water, the resulting extracted solution was concentrated with a rotary evaporator and vacuum-dried at 90C, and then passed through an alumina column (developing solvent: CH2Cl2). The extracted solution was concentrated again with a rotary evaporator and vacuum-dried at 90C so as to obtain 54.59 g of a product; the yield was 85.8%. The resulting compound was identified with a nuclear magnetic resonance analyzer (BRUKER Ultra Shield 300 NMR Spectrometer, manufactured by BRUKER Limited.). The resulting spectral data are shown below. 1H-NMR (300 MHz, solvent: CDCl3, standard substance: tetramethylsilane) delta 3.14 (m, 12H) 2.99 (m, 4H) 1.54 (m, 4H) 1.33 (m, 4H) 1.22 (t, 18H) 0.97 (t, 6H) 19F-NMR (282 MHz, solvent: CDCl3, standard substance: CF3Cl) delta -78.75 (s, 6F) 31P-NMR (121 MHz, solvent: CDCl3, standard substance: triphenylphosphine) delta 43.85 (m, 1P) The structural formula is shown below (in the formula, the dashed lines show a conjugated structure). [Show Image] The melting point was measured with a differential scanning calorimeter (DSC8230, manufactured by Shimadzu Corp.). The melting point was 25.4C. The thermal decomposition temperature was measured with a thermal gravimetry analyzer (TG8120, manufactured by Rigaku Corp.). The 5% weight-loss temperature measured at a temperature rise rate of 10C/min was 362.5C. The electrical conductivity as measured with the AC impedance method (Electrochemical Measurement System HZ-3000, manufactured by Hokuto Denko Corp.) was 0.0642 Sm-1 at 50C. The potential window was -0.1 V to 4.8 V with respect to Li/Li+, which was obtained from a cyclic voltammogram measured with the Electrochemical Measurement System HZ-3000 manufactured by Hokuto Denko Corp. using Pt for a working electrode and a counter electrode and Li for a reference electrode. A CV curve of tris(diethylamino)di-n-butylaminophosphonium bistrifluoromethane sulfonylimide is shown in FIG. 3. To 3.8 g (0.0058 mol) of tris(diethylamino)di-n-butylaminophosphonium bistrifluoromethane sulfonyl imide, an aqueous solution dissolving 5 g of NaOH in 20 ml of H2O was added, and then the resulting reaction mixture was stirred at 50C for 14 hours. Subsequently, 50 ml of CH2Cl2 were added to the reaction mixture, and the resultant solution was separated. The organic layer was washed with 30 ml of ultrapure water three times, vacuum-concentrated, and vacuum-dried at 80C so as to obtain 3.7 g of a product; the yield was 96%. A similar experiment was carried out using ethylmethylimidazolium bistrifluoromethane sulfonylimide; the yield was 81 %. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science













 HazMat Fee +
HazMat Fee +

 For Research Only
For Research Only
 120K+ Compounds
120K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping