Alternatived Products of [ 1677-46-9 ]

Product Citations
SAR study of piperidine derivatives as inhibitors of 1,4-dihydroxy-2-naph- thoate isoprenyltransferase (MenA) from Mycobacterium tuberculosis
Berg, Kaja
;
Hegde, Pooja
;
Pujari, Venugopal
, et al.
Eur. J. Med. Chem.,2023,249,115125.
DOI:
10.1016/j.ejmech.2023.115125
PubMed ID:
36682292
More
Abstract: The electron transport chain (ETC) in the cell membrane consists of a series of redox complexes that transfer electrons from electron donors to acceptors and couples this electron transfer with the transfer of protons (H+) across a membrane. This process generates proton motive force which is used to produce ATP and a myriad of other functions and is essential for the long-term survival of Mycobacterium tuberculosis (Mtb), the causative organism of tuberculosis (TB), under the hypoxic conditions present within infected granulomas. Menaquinone (MK), an important carrier molecule within the mycobacterial ETC, is synthesized de novo by a cluster of enzymes known as the classic/canonical MK biosynthetic pathway. MenA (1,4-dihydroxy-2-naphthoate prenyltransferase), the antepenultimate enzyme in this pathway, is a verified target for TB therapy. In this study, we explored structure-activity relationships of a previously discovered MenA inhibitor scaffold, seeking to improve potency and drug disposition properties. Focusing our campaign upon three molecular regions, we identified two novel inhibitors with potent activity against MenA and Mtb (IC50 = 13-22 μM, GIC50 = 8-10 μM). These analogs also displayed substantially improved pharmacokinetic parameters and potent synergy with other ETC-targeting agents, achieving nearly complete sterilization of Mtb in combination therapy within two weeks in vivo. These new inhibitors of MK biosynthesis present a promising new strategy to curb the continued spread of TB.
Keywords:
1,4-dihydroxy-2-naphthoate prenyltransferase ;
MenA ;
MenA inhibitors ;
Menaquinone ;
Mtb ;
Mycobacterium tuberculosis ;
Piperidine derivatives ;
SAR
Purchased from AmBeed:
25952-53-8 ;
90719-32-7 ;
872-85-5 ;
6457-49-4 ;
3769-41-3 ;
10338-57-5 ;
135-19-3 ;
135-19-3 ;
28177-48-2 ;
22246-18-0 ;
122334-37-6 ;
91914-06-6 ;
10040-98-9 ;
161975-39-9 ;
150-76-5 ;
371-41-5 ;
63754-96-1 ;
288-32-4 ;
3380-34-5 ;
1677-46-9 ;
166815-96-9 ;
700-57-2 ;
1204-86-0 ;
21725-69-9 ;
367-12-4 ;
1003-29-8 ;
627-35-0 ;
27292-49-5 ;
104324-16-5 ;
123855-51-6 ;
4328-13-6 ;
875401-70-0 ;
405272-71-1 ;
63614-86-8 ;
1420942-13-7 ;
25952-53-8 ;
1420895-21-1 ;
1078-18-8 ;
32363-45-4 ;
69564-68-7 ;
31519-22-9 ;
22246-18-0 ;
189618-33-5 ;
180847-24-9 ;
6264-98-8 ;
946680-75-7 ;
63608-38-8 ;
713-68-8 ;
62810-39-3 ;
189618-32-4 ;
180847-23-8 ;
63608-31-1 ;
15789-05-6 ;
63712-27-6 ;
63608-33-3 ;
63608-35-5
...More
 from Mycobacterium tuberculosis.png)
Product Details of [ 1677-46-9 ]
| CAS No. : | 1677-46-9 |
MDL No. : | MFCD00024052 |
| Formula : |
C10H9NO2
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | RTNPPPQVXREFKX-UHFFFAOYSA-N |
| M.W : |
175.18
|
Pubchem ID : | 54686436 |
| Synonyms : |
|
Application In Synthesis of [ 1677-46-9 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Downstream synthetic route of [ 1677-46-9 ]
- 1
-
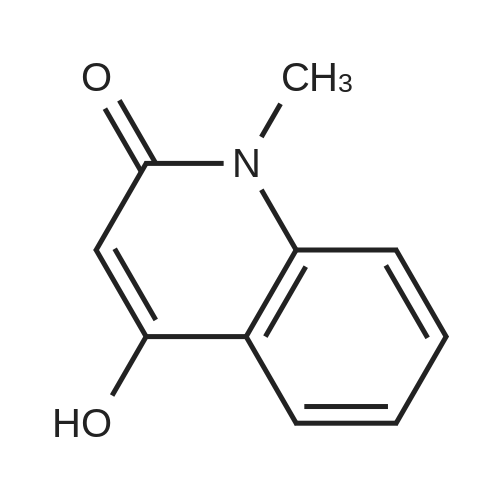 [ 1677-46-9 ]
[ 1677-46-9 ]

-
 [ 6374-91-0 ]
[ 6374-91-0 ]

-
 [ 109-77-3 ]
[ 109-77-3 ]

-
2-amino-6-methyl-5',7'-dibromo-2',5-dioxo-5,6-dihydrospiro[pyrano[3,2-c]quinoline-4,3'-indoline]-3-carbonitrile
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 87% |
With 2-amino-2-hydroxymethyl-1,3-propanediol; In ethanol; at 20℃; for 3.5h; |
General procedure: To a well-stirred solution of isatin and malononitrile(1 mmol each) in ethanol (95%, 4 mL) was added dimedone,4-hydroxycoumarin, 4-hydroxy-N-methylquinolin-2-one,or 2-methyl-pyrazol-2-one [generated in situ from ethylacetoacetate and hydrazine hydrate, 1 mmol each]. To thissolution was added THAM (30 mol %) and stirring wascontinued at ambient temperature. Upon completion of thereaction (TLC), water (5 mL) was added and stirring wascontinued for 10 min more. Resultant solid product wasfiltered, washed repeatedly with water, and dried. The dried solid was washed thrice with hexaneechloroformmixture (1:1, v/v) and dried again. Resultant product didnot require any further purification. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 120K+ Compounds
120K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping


 from Mycobacterium tuberculosis.png)





