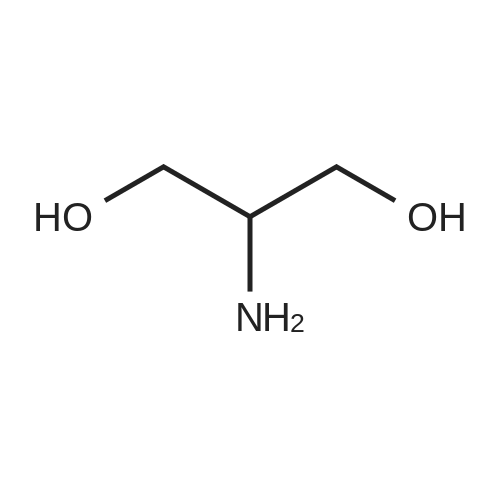
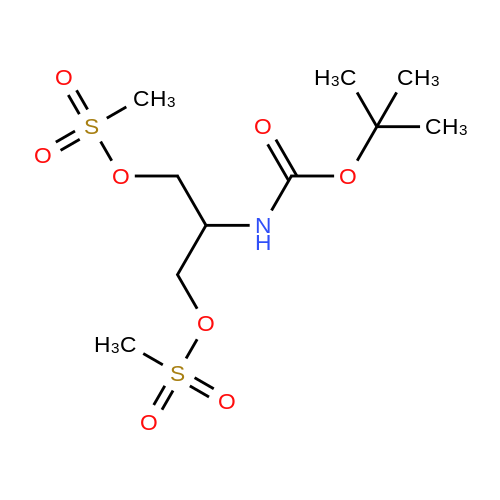
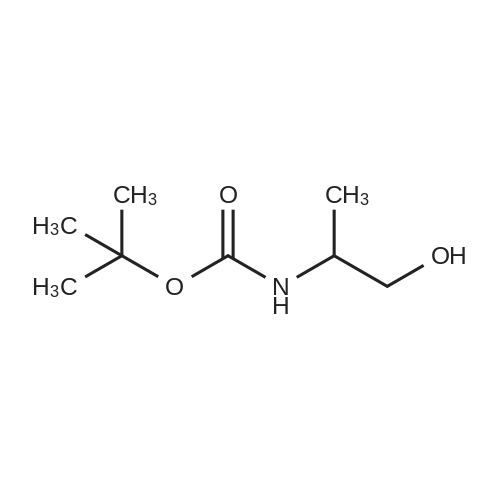
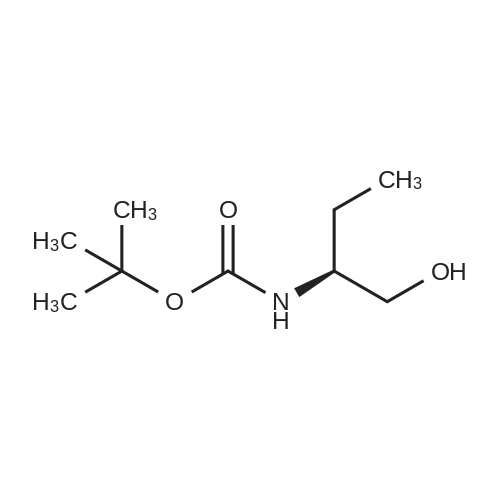
| 95% |
With trimethylamine; In dichloromethane; for 4h; |
Serinoi (2,00 g, 22,0 mmol) was dissolved in dichioromethane (40 mL) and trimethylamitie (10 mL). Di-tert-butyl dicarbonate (5.76 g, 26.4 mmol, 1.2 eq) was then added, and the reaction stirred for 4 hours. The mixture was evaporated and the residue portioned between ethyl acetate and water. The organic fraction was washed with water ( lx), 1M HC1 (2x), saturated sodium bicarbonate (lx), and brine (lx) before drying over sodium sulfate and evaporation to give compound 23 (3.99 g, 20.9 mmol) in 95% yield, which was used without purification in further steps. |
| 91% |
In ethanol; at 20℃; for 6h; |
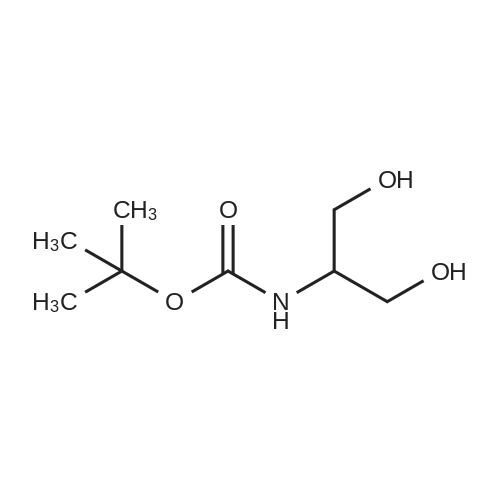
2-amino-1,3-propanediol (5 g) was dissolved in 150 mL of absolute ethanol, A solution of 100 mL of Boc anhydride (12 g) in absolute ethanol was added, Room temperature reaction 6h, Remove the solvent, Recrystallization from n-heptane gave a white solid 3 (9.6 g, 91%). |
| 72% |
With N-ethyl-N,N-diisopropylamine; In methanol; for 0.25h;Product distribution / selectivity; |
Example 2Lambda/-Boc-Serinol. Lambda/,Lambda/-diisopropylethylamine (DIEA; 10.3 ml_, 59 mmol, 1.1 equiv) was added to serinol (4.88 g, 54 mmol, 1 equiv) in 10 ml_ methanol. A solution of di-f-butyl dicarbonate (12.9 g, 59 mmol, 1.1 equiv.) in 5 mL methanol was then added to the original solution. The reaction was exothermic and evolved gas. The reaction appeared to be complete after 15 min. (as determined by TLC analysis in 9:1 CHCI3:CH3OH with ninhydrin stain). The solvent was then removed at reduced pressure, and the crude material was purified by flash chromatography, using CHCI3:CH3OH (first at a 9:1 ratio and then at a 85:15 ratio), to yield 7.18g (72% yield) of white, flaked crystals. [Rf: 0.35 (5:1 CHCI3:CH3OH); melting point 85-860C; 1H NMR (200 MHz, CDCI3) delta 5.25 (br d, J = 5.6, 1 H, NH), 3.9-3.65 (m, 5H, (-CH2)2CH-), 2.62 (t, J = 5.6, 2H, OH), 1.45 (s, 9H, C(CHa)3)]. EPO <DP n="30"/>Example 3[0107] To prepare the bis-phthalonitrile used in scheme 2, the nitrogen of serinol was Boc-protected, and the serinol was then reacted with 3-nitrophthalonitrile on a multi-gram scale, to give the desired tethered bis-phthalonitrile, in 70% yield |
| 62% |
With potassium carbonate; In tetrahydrofuran; water; at 20℃; |
2-Amino-1,3-propanediol (1.62 g, 17.8 mmol, 1.0 eq) was dissolved in 60 mL of tetrahydrofuran/H2O (1/1). Di-tert-butyl dicarbonate (Boc2O) (5.82 g, 26.7 mmol, 1.5 eq) and K2CO3 (6.15 g, 44.5 mmol,2.5 eq) were added. The reaction was stirred at room temperature overnight. The reaction systemwas allowed to stand for stratification, and the aqueous phase was extracted three times withtetrahydrofuran. The organic phase was combined, dried over anhydrous Na2SO4, evaporatedin vacuo, and puried by column chromatography (silica gel:petroleum ether/ethyl acetate, 1:1) toproduce compound 4 as a white solid in a 62% yield (2.1 g, 11.0 mmol). 1H-NMR (400 MHz, CDCl3) 5.24 (s, 1H), 3.81 (qd, J = 11.1, 4.3 Hz, 4H), 3.69 (s, 1H), 2.50 (dd, J = 22.1, 14.7 Hz, 2H), 1.46 (s, 9H),which agrees with published data [39]. |
| 49% |
With triethylamine; In tetrahydrofuran; at 0 - 20℃; |
2-Amino-1 ,3-propanediol (5.Og, 54.9mmol) was dissolved in dry THF (175ml) and triethylamine (7.7ml) added. The solution was cooled in an ice-bath and di-fe/f- butylcarbonate (11.98g, 54.9mmol) added in portions over 15mins. The solution was allowed to warm to ambient temperature and stirred for 90mins. The solvent was evaporated and water (250ml) added and the product extracted into ethyl acetate (4 x 125ml). The combined organics were washed with brine, dried over magnesium sulphate, filtered and evaporated. The product was isolated by recrystallization from hot ethyl acetate-petrol (1 :3) to give shiny flakes 5.18g (49% yield). The structure was confirmed by 1H NMR (300MHz , CDCI3): 1.44 (s, 9H), 3.08-3.17 (m, 1 H), 3.61 - 3.84 (m, 4H). |
|
With triethylamine; In dichloromethane; at 20℃;Cooling with ice; |
General procedure: Diols or diamine (0.048 mol) and triethylamine (4.81 g, 0.057 mol) were dissolved in anhydrous CH2Cl2 (50 mL). (Boc)2O (12.45 g, 0.057 mol) in anhydrous CH2Cl2 solution was added dropwise to the above stirred solution under the ice bath. The mixture was stirred overnight at room temperature, followed by evaporation of the organic solvents. The residue was purified with silica gel column chromatography (dichloromethane/methyl alcohol = 30: 1). The synthesis of naked two primary amine compound according to the literature [29]. Then, acryloyl chloride (6.95 g, 0.077 mol) in anhydrous dichloromethane (50 mL) was added dropwise to a stirred solution of diol or diamine (0.038 mol) and triethylamine (7.77 g, 0.077 mol) in anhydrous dichloromethane (50 mL) under the ice bath. The mixture was stirred overnight at room temperature and then filtered off generated salt, followed by evaporation of the volatile solvent. The residue was purified with silica gel column chromatography (PE: EA = 3: 1, v/v) to give LC1-LC6. |
|
With triethylamine; In dichloromethane; at 0 - 20℃; |
General procedure: Diethanolamine 3 (or threoninol 5, 3-amino-1,2-propanediol 6)(0.047 mol) was dissolved in CH2Cl2 and triethylamine (9.60 g,0.095 mol) was slowly added. The solution was cooled to 0 C,and (Boc)2O (12.45 g, 0.057 mol) was added dropwise. Then themixed solution was left for stirring at room temperature overnight.The solvent was removed under reduce pressure to afford crudeproduct, which was further purified by silica gel column chromatography(CH2Cl2/CH3OH = 10/1, v/v) to give compound 3a (or5a, 6a). |
|
With triethylamine; In dichloromethane; |
Referring to Scheme LLL, the amino group in diol 1-29 is protected with Boc2O to provide compound 1-30 which is mesylated (e.g., with MsC1 and TEA) to afford compound 1-31. Compound 1-31 is subjected to a double nucleophilic substitution with pyrazolone 1-9, providing1-32, which is brominated (e.g., with NBS) to afford compound 1-33. Sequential treatment of 1-33 with n-butyl lithium, zinc chloride, and TCPC provides compound 1-34. Intermediate 61 is obtained when 1-34 is reacted with aqueous ammonia. |
|
In methanol; water; at 0 - 25℃; for 4.5h; |
Add 6mL of deionized water and 14mL of methanol to the three-necked flask, stir and mix well, then add 0.02mol serinol to the mixed solution, stir and dissolve it completely, cool to 0 C with an ice bath, and then add the three mixed solutions Slowly add 0.022mol of di-tert-butyl dicarbonate. During the dropwise addition, the temperature was maintained at 0 C. After 30 minutes, the ice bath was removed and the reaction was performed at a constant 25 C for 4 hours. After the reaction was completed, extraction was performed three times with dichloromethane. The solvent methanol and water were distilled off under reduced pressure, and vacuum-dried at 45 C. for 24 h to obtain the product tert-butyl 1,3-dihydroxypropane-2-carbamate. |
|
With triethylamine; In dichloromethane; for 4h; |
Serinol (2.00 g, 22.0 mmol) was dissolved in dichloromethane (40 mL) and trimethyamine ( 10 mL). Di-tert-butyl dicarbonate (5.76 g, 26.4 mmol, 1 .2 eq) was then added, and the reaction stirred for 4 hours. The mixture was evaporated and the residue portioned between ethyl acetate and water. The organic fraction was washed with water ( l x), 1 M HCI (2x), saturated sodium bicarbonate ( l x), and brine ( l x) before drying over sodium sulfate and evaporation to give compound 23 (3.99 g, 20.9 mmol) in 95% yield, which was used without purification in further steps. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 120K+ Compounds
120K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping