| Identification | Back Directory | [Name]
DIAZOXIDE | [CAS]
364-98-7 | [Synonyms]
Eudemine
Sch-6783
mutabase
srg95213
dizoxide
diazoxid
Proglycem
DIAZOXIDE
proglicem
hyperstat
NSC-64198
Diazoxided
diazossido
Diazoxide CRS
Diazoxide API
DIAZOXIDE,USP
Diazoxide-13CD3
Diazoxide (200 mg)
Diazoxide (1186000)
DIAZOXIDE USP/EP/BP
Desipramine Impurity 7
DIAZOXIDE ACTIVATOR OF ATP-DEPE
Diazoxide Benzothiadiazinone Analog
DIAZOXIDE 364-98-7 kf-wang(at)kf-chem.com
7-Chloro-3-methyl-2H-1,2,4-benzothiadiazine1
3-Methyl-7-chloro-1,2,4-benzothiadiazine 1,1-dioxide
7-cloro-3-metil-2h-1,2,4-benzotiodiazina-1,1-diossido
7-chloro-3-methyl-4H-1λ,2,4-benzothiadiazine-1,1-dione
7-CHLORO-3-METHYL-2H-1,2,4-BENZOTHIADIAZINE 1,1-DIOXIDE
2h-1,2,4-benzothiadiazine,7-chloro-3-methyl-,1,1-dioxide
7-chloro-3-methyl-4h-1λ6,2,4-benzothiadiazine 1,1-dioxide
7-chloro-3-methyl-4H-benzo[e][1,2,4]thiadiazine 1,1-dioxide
Diazoxide,7-Chloro-3-methyl-2H-1,2,4-benzothiadiazine 1,1-dioxide
Diazoxide impurity-4 (7-chloro-3-methyl-2H-1,2,4-benzothiadiazine 1,1-dioxide) | [EINECS(EC#)]
206-668-1 | [Molecular Formula]
C8H7ClN2O2S | [MDL Number]
MFCD00078578 | [MOL File]
364-98-7.mol | [Molecular Weight]
230.67 |
| Chemical Properties | Back Directory | [Melting point ]
>310°C | [Boiling point ]
414.8±47.0 °C(Predicted) | [density ]
1.3767 (rough estimate) | [refractive index ]
1.6300 (estimate) | [storage temp. ]
Store at RT | [solubility ]
0.1 M NaOH: soluble
| [form ]
neat | [pka]
pKa 8.5 (Uncertain) | [color ]
White | [Water Solubility ]
Soluble in 0.1M NaOH. Insoluble in water or in methanol. | [λmax]
268nm(MeOH)(lit.) | [Merck ]
14,3004 | [Stability:]
Stable for 2 years from date of purchase as supplied. Solutions in DMSO may be stored at -20° for up to 3 months. |
| Hazard Information | Back Directory | [Description]
Diazoxide is a nondiuretic derivative of thiazides that dramatically reduces blood pressure
by direct relaxation of smooth muscles of the arterioles, possibly as a result of calcium channel activation of smooth musculature in arterioles. It has a weak effect on the venous
system and on the heart. In addition to hypotensive action, diazoxide causes a sharp increase
in the level of glucose in the blood as a result of the inhibition of insulin release from adrenal
glands. Some of the undesirable effects are water and sodium ion retention in the body
and increased concentrations of uric acid in the blood. It is used in urgent situations where
blood pressure needs to be reduced in severe hypertension. Diazoxide is not used for essential
hypertension. A synonym of this drug is hyperstat. | [Chemical Properties]
White Solid | [Originator]
Eudemine,Allen and
Hanburys,UK,1970 | [Uses]
Diazoxide reduces status epilepticus neuron damage in diabetes. | [Uses]
wound healing agent | [Definition]
ChEBI: A benzothiadiazine that is the S,S-dioxide of 2H-1,2,4-benzothiadiazine which is substituted at position 3 by a methyl group and at position 7 by chlorine. A peripheral vasodilator, it increases the
oncentration of glucose in the plasma and inhibits the secretion of insulin by the beta- cells of the pancreas. It is used orally in the management of intractable hypoglycaemia and intravenously in the management of hypertensive emergencies. | [Brand name]
Hyperstat (Schering); Proglycem (Baker Norton). | [Therapeutic Function]
Antihypertensive
| [Biological Functions]
Diazoxide (Hyperstat) is chemically similar to the thiazide
diuretics. It is devoid of diuretic activity and
causes Na+ and water retention. Diazoxide is a very potent
vasodilator and is available only for intravenous use in the treatment of hypertensive emergencies. The
mechanism by which diazoxide relaxes vascular smooth
muscle is related to its ability to activate potassium
channels and produce a hyperpolarization of the cell
membrane. | [General Description]
Diazoxide is 7-chloro-3-methyl-4H-benzo[e][1,2,4]thiadiazine-1,1-dioxide , and is currentlyavailable in the United States only as a 50-mg/mLoral suspension (Proglycem); discontinued formulations includedcapsules for oral administration, and injectable formsthat typically found use for indications other than hypoglycemicconditions. Diazoxide is a cyclic benzenesulfonamide,although the free acid in solution can exist in threetautomeric forms, and the 4H tautomer most likely predominatesto a very high proportion. Partly because of theadditional nitrogen in the quinazoline ring structure, themolecule is somewhat more acidic (pKa~8.4, 8.6)than benzenesulfonamide (pKa~10). | [General Description]
Diazoxide is used as the sodium salt of7-chloro-3-methyl-2H-1,2,4-benzothiadiazine 1,1-dioxide(Hyperstat IV). Diazoxide lowers peripheral vascular resistance,increases cardiac output, and does not compromiserenal blood flow.
This is a des-sulfamoyl analog of the benzothiazine diureticsand has a close structural similarity to chlorothiazide. Itwas developed intentionally to increase the antihypertensiveaction of the thiazides and to minimize the diuretic effect. | [Biological Activity]
Antihypertensive, activates ATP-dependent K + channels. Induces activation of PKC ε , an intermediate in the opening of mitoK ATP channels, results in cardioprotection against hypoxia-induced death. Blocks desensitization of AMPA receptors. | [Biochem/physiol Actions]
Selective ATP-sensitive K+ channel activator in both vascular smooth muscle and pancreatic β-cells; antihypertensive. | [Mechanism of action]
Diazoxide reduces peripheral vascular resistance and blood pressure by a direct vasodilating effect on the VSM with a
mechanism similar to that described for minoxidil by activating (opening) the ATP-modulated potassium channel.
Thus, diazoxide prolongs the opening of the potassium channel, sustaining greater vasodilation on arterioles than on
veins. The greatest hypotensive effect is observed in patients with malignant hypertension. Although oral or slow IV
administration of diazoxide can produce a sustained fall in blood pressure, rapid IV administration is required for
maximum hypotensive effects, especially in patients with malignant hypertension. Diazoxide-induced decreases in
blood pressure and peripheral vascular resistance are accompanied by a reflex response, resulting in an increased
heart rate, cardiac output, and left ventricular ejection rate. In contrast to the thiazide diuretics, diazoxide causes
sodium and water retention and decreased urinary output, which can result in expansion of plasma and extracellular
fluid volume, edema, and congestive heart failure, especially during prolonged administration.
Diazoxide increases blood glucose concentration (diazoxide-induced hyperglycemia) by several different mechanisms:
by inhibiting pancreatic insulin secretion, by stimulating release of catecholamines, or by increasing hepatic release of
glucose. The precise mechanism of inhibition of insulin release has not been elucidated but, possibly, may result
from an effect of diazoxide on cell-membrane potassium channels and calcium flux. | [Pharmacokinetics]
Following rapid IV administration, diazoxide produces a prompt reduction in blood pressure, with maximum hypotensive
effects occurring within 5 minutes. The duration of its hypotensive effect varies from 3 to 12 hours, but ranges from 30
minutes to 72 hours have been observed. The elimination half-life of diazoxide following a single oral or IV dose has
been reported to range from 21 to 45 hours in adults with normal renal function. In patients with renal impairment, the
half-life is prolonged. Approximately 90% of the diazoxide in the blood is bound to plasma proteins. Approximately 20 to
50% of diazoxide is eliminated unchanged in the urine, along with its major metabolites, resulting from the oxidation of
the 3-methyl group to its 3-hydroxymethyl- and 3-carboxyl-metabolites. | [Pharmacology]
The hemodynamic effects of diazoxide are similar to
those of hydralazine and minoxidil. It produces direct
relaxation of arteriolar smooth muscle with little effect
on capacitance beds. Since it does not impair cardiovascular
reflexes, orthostasis is not a problem. Its administration
is, however, associated with a reflex increase in
cardiac output that partially counters its antihypertensive
effects. Propranolol and other �-blockers potentiate
the vasodilating properties of the drug. Diazoxide
has no direct action on the heart. Although renal blood
flow and glomerular filtration may fall transiently, they
generally return to predrug levels within an hour. | [Clinical Use]
Diazoxide is used by intravenous injection as a rapidly acting antihypertensiveagent for emergency reduction of blood pressurein hospitalized patients with accelerated or malignanthypertension. More than 90% is bound to serum protein, andcaution is needed when it is used in conjunction with otherprotein-bound drugs that may be displaced by diazoxide.The injection is given rapidly by the intravenous route toensure maximal effect. The initial dose is usually 1 mg/kg ofbody weight, with a second dose given if the first injectiondoes not lower blood pressure satisfactorily within 30 minutes.Further doses may be given at 4- to 24-hour intervalsif needed. Oral antihypertensive therapy is begun as soon aspossible. | [Clinical Use]
In contrast to the acute clinical uses of glucagon, diazoxidefinds use in chronic hypoglycemic conditions: inoperableislet cell adenoma or carcinomas, extrapancreatic malignanciesof insulin-secreting cells, or islet cell hyperplasias. Inchildren, additional indications include congenital hyperinsulinemia124and leucine sensitivity. Experimentally, diazoxideis among an array of ATP-sensitive potassium channel openersbeing studied for intermittently bringing aboutβ-cell rest. | [Side effects]
Since diazoxide is not often used for long-term treatment,
toxicities associated with chronic use are rare.The
chief concern is the side effects associated with the increased
workload on the heart, which may precipitate
myocardial ischemia and Na+ and water retention.
These undesirable effects can be controlled by concurrent
therapy with a β-blocker and a diuretic.
Diazoxide may cause hyperglycemia, especially in
diabetics, so if the drug is used for several days, blood
glucose levels should be measured.
When used in the treatment of toxemia, diazoxide
may stop labor, because it relaxes uterine smooth muscle. | [Synthesis]
7-chloro-3-methyl-2-H-1,2,4-benzothiadiazin-1,1-dioxide (21.3.14), is synthesized
by condensating 2-aminosulfonyl-4-chloroaniline with triethyl orthoacetate.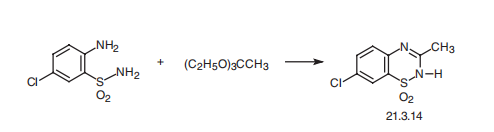
| [Veterinary Drugs and Treatments]
Oral diazoxide is used in canine and ferret medicine for the treatment
of hypoglycemia secondary
to hyperinsulin secretion (e.g., insulinoma).
Insulinomas are apparently very rare in the cat; there is
little experience with this drug in that species.
In human medicine, intravenous diazoxide is sometimes used for
treating severe hypertension. | [Drug interactions]
Potentially hazardous interactions with other drugs
Antihypertensives and vasodilators: enhanced
hypotensive effect.
MAOIs: withdraw at least 14 days before starting
diazoxide
Phenytoin: may reduce phenytoin levels. | [Environmental Fate]
Diazoxide is a potassium channel activator, which causes local
relaxation in smooth muscles by increasing membrane
permeability to potassium ions. Consequently, voltage-gated
calcium ion channels are ineffective, inhibiting the generation
of an action potential. The primary mechanism by which
diazoxide lowers blood pressure is by direct relaxation of
medium sized blood vessels. The cardiac output and renin
secretion increases, resulting in elevated angiotensin II levels
and retention of salt and water. When used to treat low blood
sugar, diazoxide decreases insulin release from the pancreas. | [Metabolism]
Diazoxide lowers blood pressure within 3 to 5 minutes
after rapid intravenous injection, and its duration of action
may be 4 to 12 hours. Interestingly, if diazoxide is
either injected slowly or infused its hypotensive action
is quite modest.This is believed to be due to a rapid and
extensive binding of the drug to plasma proteins. Both
the liver and kidney contribute to its metabolism and
excretion.The plasma half-life is therefore prolonged in
patients with chronic renal failure. | [storage]
Room temperature | [References]
1) Trube et al. (1986), Opposite effects of tolbutamide and diazoxide on the ATP-dependent K+ channel in mouse pancreatic beta-cells.l; Pflugers Arch., 407 493
2) Kim et al. (2006), Diazoxide acts more as a PKC-epsilon activator, and indirectly activates the mitochondrial K(ATP) channel conferring cardioprotection against hypoxic injury; Br. J. Pharmacol, 149 1059
3) Coetzee (2013), Multiplicity of effectors of the cardioprotective agent, diazoxide; Pharmacol. Ther., 140 167
4) Jasova et al. (2016), Stimulation of mitochondrial ATP synthase activity – a new diazoxide-mediated mechanism of cardioprotection; Physiol. Res., 65 Suppl 1 S119
5) Salgado-Puga et al. (2017), Subclinical Doses of ATP-sensitive Potassium channel Modulators Prevent Alterations in Memory and Synaptic Plasticity Induced by Amyloid-β; J. Alzheimers Dis., 57 205 |
|
|