| Identification | Back Directory | [Name]
ABT-199 | [CAS]
1257044-40-8 | [Synonyms]
GDC0199
ABT-199
GDC 0199
GDC-0199
Venetoclax
ABT199,GDC0199
2-(1H-pyrrolo[2,3-b]pyridin-5-yloxy)-4-(4-((2-(4-chlorophenyl)-4,4-dimethylcyclohex-1-enyl)methyl)piperazin-1-yl)-N-(3-nitro-4-((tetrahydro-2H-pyran-4-yl)methylamino)phenylsulfonyl)benzamide
4-[4-[[2-(4-Chlorophenyl)-4,4-dimethyl-1-cyclohexen-1-yl]methyl]-1-piperazinyl]-N-[[3-nitro-4-[[(tetrahydro-2H-pyran-4-yl)methyl]amino]phenyl]sulfonyl]-2-(1H-pyrrolo[2,3-b]pyridin-5-yloxy)-benzamide
Benzamide, 4-[4-[[2-(4-chlorophenyl)-4,4-dimethyl-1-cyclohexen-1-yl]methyl]-1-piperazinyl]-N-[[3-nitro-4-[[(tetrahydro-2H-pyran-4-yl)methyl]amino]phenyl]sulfonyl]-2-(1H-pyrrolo[2,3-b]pyridin-5-yloxy)-
2-((1H-Pyrrolo[2,3-b]pyridin-5-yl)oxy)-4-(4-((4'-chloro-5,5-dimethyl-3,4,5,6-tetrahydro-[1,1'-biphenyl]-2-yl)methyl)piperazin-1-yl)-N-((3-nitro-4-(((tetrahydro-2H-pyran-4-yl)methyl)amino)phenyl)sulfonyl)benzamide
ABT 199
4-[4-[[2-(4-Chlorophenyl)-4,4-dimethyl-1-cyclohexen-1-yl]methyl]-1-piperazinyl]-N-[[3-nitro-4-[[(tetrahydro-2H-pyran-4-yl)methyl]amino]phenyl]sulfonyl]-2-(1H-pyrrolo[2,3-b]pyridin-5-yloxy)-benzamide
4-[4-[[2-(4-Chlorophenyl)-4,4-dimethyl-1-cyclohexen-1-yl]methyl]-1-piperazinyl]-N-[[3-nitro-4-[[(tetrahydro-2H-pyran-4-yl)methyl]amino]phenyl]sulfonyl]-2-(1H-pyrrolo[2,3-b]pyridin-5-yloxy)-benzamide ABT-199 (GDC-0199) | [EINECS(EC#)]
820-130-9 | [Molecular Formula]
C45H50ClN7O7S | [MDL Number]
MFCD23160052 | [MOL File]
1257044-40-8.mol | [Molecular Weight]
868.439 |
| Chemical Properties | Back Directory | [Melting point ]
>150°C (dec.) | [density ]
1.340±0.06 g/cm3(Predicted) | [storage temp. ]
-20°C Freezer | [solubility ]
DMSO (Slightly) | [form ]
Yellow solid. | [pka]
4.09±0.10(Predicted) | [color ]
Light Yellow to Yellow | [InChIKey]
LQBVNQSMGBZMKD-UHFFFAOYSA-N | [SMILES]
C(NS(C1=CC=C(NCC2CCOCC2)C([N+]([O-])=O)=C1)(=O)=O)(=O)C1=CC=C(N2CCN(CC3CCC(C)(C)CC=3C3=CC=C(Cl)C=C3)CC2)C=C1OC1=CN=C2NC=CC2=C1 |
| Questions And Answer | Back Directory | [Description]
ABT-199(1257044-40-8), also known as venetoclax, is a small, oral chemical molecule used for the treatment of chronic lymphocytic leukemia (CLL) associated with a specific chromosomal abnormality. As a second line treatment drug of CLL, ABT-199 take effects through acting as a BH3-mimetic and inhibitor of Bcl-2 (the anti-apoptotic B-cell lymphoma-2protein), further inducing apoptosis of CLL cells but not platelets. It also has the potential for the treatment of ER-positive breast cancer.
| [BCL-2 inhibitor]
Venetoclax(1257044-40-8) is a first-in-class, oral, selective BCL-2 inhibitor (BH3-only mimetic). The drug is approved in numerous countries, including those of the EU and in the USA, for the treatment of adults with relapsed or refractory (RR) CLL. The drug arose from research by Abbott Laboratories (now AbbVie) during a collaboration with Genentech and is being codeveloped by AbbVie and Genentech/Roche primarily.
| [Mechanism of action]
Venetoclax(1257044-40-8) has high affinity for BCL-2, binding to the protein with an affinity more than three orders of magnitude greater than to BCL-XL or BCL-W in vitro. By binding to BCL-2, the drug displaces BCL-2-bound proapoptotic proteins (such as BIM), resulting in the permeabilization of mitochondrial outer membranes, activation of caspases, and restoration of cancer cell apoptosis, with this process requiring the apoptosis regulators BAX or BAK.
| [Clinical evaluation]
As a specific Bcl-2 inhibitor of Bcl-2, ABT-199 was approved by FDA to treat CLL in 2015. ABT-199 was designed to avoid the nonselective binding of ABT-263 with Bcl-XL, which could induce the adverse effect of thrombocytopenia. The X-ray crystal structure of a Bcl-2 dimer in complex with ABT-263 and that with a modified ABT-263 lacking the thiophenyl moiety elucidated an alternative bind site in the hotspot p4-binding pocket that could be utilized for selective binding of Bcl-2. This led to the development of ABT-199 that tightly binds to Bcl-2 but not Bcl-XL. Consistent with its lower affinity to Bcl-XL, ABT-199 spared human platelets both in vivo and in vitro. In clinical trials, ABT-199 exhibited immediate antileukemic activity after a single dose in three refractory CLL patients with only minor changes in platelet counts. ABT-199 as a single agent showed promising clinical response in many malignancies, such as diffused large B-cell lymphoma, follicular lymphoma, CLL, acute myeloblastic leukemia (AML), and multiple myeloma. Studies also showed that ABT-199 had a significant sensitizing effect in combination with other antitumor drugs, such as rituximab, obinutuzumab, in CLL and AML patients. However, ABT-199 did show adverse effects, especially tumor lysis syndrome (TLS). Furthermore, neutropenia or infections have also been observed in patients using ABT-199.
Chapter 10 - Bcl-2 Inhibitors as Sensitizing Agents for Cancer Chemotherapy | [Cytotoxic Activity]
Venetoclax displayed cytotoxic activity in various tumour samples/cell lines, including some derived from CLLs, various other NHL subtypes, acute lymphoblastic leukaemias (ALLs), AMLs, chronic myeloid leukaemias (CMLs) and MMs. Notably, the sensitivity of venetoclax correlated with higher BCL-2 expression, with a BCL-2 high status [i.e. BCL-2 gains, BCL-2 amplifications, or the t translocation, which is a notable cause of deregulated BCL-2 expression] and higher BCL-2/MCL-1 ratios being potentially predictive of sensitivity to the drug.
| [References]
https://en.wikipedia.org/wiki/Venetoclax
Vogler, M, et al. "ABT-199 selectively inhibits BCL2 but not BCL2L1 and efficiently induces apoptosis of chronic lymphocytic leukaemic cells but not platelets." British Journal of Haematology 163.1(2013):139-42.
Vaillant, François, et al. "Targeting BCL-2 with the BH3 Mimetic ABT-199 in Estrogen Receptor-Positive Breast Cancer." Cancer Cell 24.1(2013):120-9.
Lindeman, G. J., et al. "Abstract P2-09-01: Targeting BCL-2 with the BH3 mimetic ABT-199 in ER-positive breast cancer." Cancer Research 73.24 Supplement (2013):P2-09-01-P2-09-01. |
| Hazard Information | Back Directory | [Uses]
ABT 199 (>99%) is a potent and selective BCL-2 inhibitor that achieves potent antitumour activity while sparing platelets. It’s practical application is to treat chronic lymphocytic leukaemic cells and estrogen receptor-positive breast cancer. | [Definition]
ChEBI: A member of the class of pyrrolopyridines that is a potent inhibitor of the antiapoptotic protein B-cell lymphoma 2. It is used for treamtment of chronic lymphocytic leukemia with 17p deletion. | [Clinical Use]
Selective inhibitor of B-cell lymphoma protein:
Treatment of chronic lymphocytic leukaemia | [Synthesis]
The manufacturing route to venetoclax takes place by
coupling of three key structural subunits: azaindole 162,
sulfonamide 165, and piperazine 172. The first of these
subunits was generated in two steps from commercially
available 4-bromo-2-fluoro-1-iodo-benzene (159).
Grignard formation of iodide 159 (i-PrMgCl) followed by
quenching with Boc2O provided the desired tert-butyl ester 160
without the need for chromatographic purification. Aromatic
substitution of crude 160 with azaindole 161 provided access to
162 in 86% yield after recrystallization from EtOAc/heptane. Sulfonamide 165 was
formed in 91% yield and 99.9% purity via aromatic substitution
of commercially available 163 with amine 164 at 80 °C
(DIPEA, MeCN).

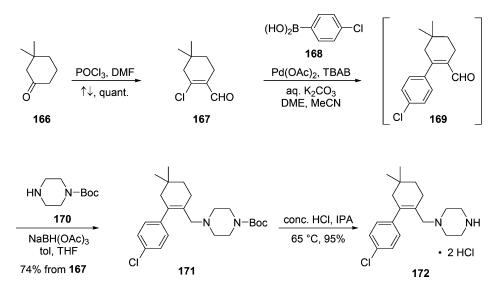
Synthesis of the third venetoclax subunit, piperazine amine
hydrochloride salt 172, began with commercial cyclohexanone
166. Vilsmeier-Haack formylation of the
sterically more accessible enol tautomer of 166 delivered vinyl
chloride 167 in quantitative yield. Coupling of this chloride
with commercial aryl boronate 168 gave rise to transient enal
169 in 87% assay yield, which was not isolated. Crude 169 was
then carried into a reductive amination reaction with
commercial N-Boc piperazine (170). Precipitation and
recrystallization from acetonitrile ultimately furnished piperazinyl
alkene 171 in 74% yield from 167. Finally, subunit 172
was obtained via Boc removal with concentrated HCl in IPA at
65 °C and subsequent filtration, conditions that provided a 95%
yield of high purity intermediate 172 (>99.5%).
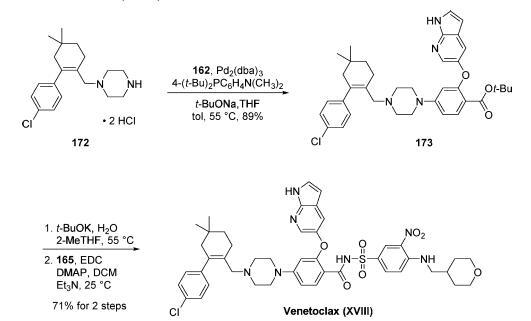
The final approach to venetoclax involved a palladiumcatalyzed
coupling of amine 172 with aryl bromide 162, ester
hydrolysis, and coupling of the resulting carboxylic acid with
sulfonamide 165. In practice, Buchwald-Hartwig amination of 162 with 172 proceeded smoothly and
relied upon workup with cysteine to enable cleansing of
residual palladium from the reaction mixture. This reaction
gave rise to advanced intermediate 173 in 89% yield after
crystallization from cyclohexane. Treatment of 173 with t-
BuOK/H2O/2-MeTHF at 55 °C provided the corresponding
free acid, which was immediately activated with EDC/DMAP/
Et3N to promote coupling with sulfonamide 165 at room
temperature. The final drug target could be accessed by
crystallization from EtOAc and washing with 1:1 DCM/EtOAc,
yielding venetoclax (XVIII) in free base form in 71% over
the two final steps. This synthetic route was capable of
fashioning the drug target in 52% overall yield based on the
longest linear sequence (7 steps).
| [Drug interactions]
Potentially hazardous interactions with other drugs
Antibacterials: concentration possibly increased by
ciprofloxacin, clarithromycin and erythromycin -
reduce venetoclax dose; avoid with rifampicin.
Anticoagulants: avoid with dabigatran; concentration
of warfarin increased.
Antidepressants: avoid with St John’s wort.
Antiepileptics: concentration possibly reduced by
carbamazepine, fosphenytoin and phenytoin - avoid.
Antifungals: concentration possibly increased by
fluconazole, itraconazole, ketoconazole, posaconazole
and voriconazole - reduce venetoclax dose.
Antipsychotics: increased risk of agranulocytosis
with clozapine - avoid.
Antivirals: concentration possibly reduced by
efavirenz and etravirine - avoid; concentration
possibly increased by ritonavir - reduce venetoclax
dose.
Bosentan: concentration of venetoclax possibly
reduced by bosentan - avoid.
Calcium channel blockers: concentration possibly
increased by diltiazem and verapamil - reduce
venetoclax dose.
Cardiac glycosides: avoid with digoxin.
Cytotoxics: avoid with everolimus.
Grapefruit juice: avoid concomitant use.
Modafinil: concentration of venetoclax possibly
reduced - avoid.
Sirolimus: avoid concomitant use.
Vaccines: avoid with live vaccines. | [Metabolism]
In vitro studies show that venetoclax is mainly
metabolised by cytochrome P450 CYP3A4. M27 was
identified as a major metabolite in plasma with an
inhibitory activity against BCL-2 that is at least 58-fold
lower than venetoclax in vitro.
Excretion is mainly by the faecal route (>99.9
%; 20.8
%
unchanged). | [storage]
Store at -20°C |
|
|