| Identification | Back Directory | [Name]
Sulfuric acid Mono-[7-oxo-2-(piperidin-4-ylcarbaMoyl)-1,6-diaza-bicyclo[3.2.1]oct-6-yl] ester | [CAS]
1174018-99-5 | [Synonyms]
MK7655
CS-2637
RELEBACTAM
Relebactam(MK-7655)
MK-7655(Relebactam)
MK-7655;MK 7655;MK7655
(2S,5R)-7-oxo-2-(piperidin-1-ium-4-ylcarbamoyl)-1,6-diazabicyclo[3.2.1]octan-6-yl sulfate
Sulfuric acid Mono-[7-oxo-2-(piperidin-4-ylcarbaMoyl)-1,6-diaza-bicyclo[3.2.1]oct-6-yl] ester
(2S,5R)-sulfuric acid mono{[(4-aminopiperidin-4-yl)carbonyl]-7-oxo-1,6-diazabicyclo[3.2.1]oct-6-yl}ester
Sulfuric acid mono[(1R,2S,5R)-7-oxo-2-[(4-piperidinylamino)carbonyl]-1,6-diazabicyclo[3.2.1]oct-6-yl] ester | [Molecular Formula]
C12H20N4O6S | [MDL Number]
MFCD28502833 | [MOL File]
1174018-99-5.mol | [Molecular Weight]
348.38 |
| Chemical Properties | Back Directory | [Melting point ]
>252°C (dec.) | [density ]
1.59 | [storage temp. ]
Hygroscopic, -20°C Freezer, Under inert atmosphere | [solubility ]
DMSO (Slightly), Water (Slightly) | [form ]
Solid | [pka]
-4.59±0.18(Predicted) | [color ]
Off-White to Light Yellow | [Stability:]
Hygroscopic | [InChI]
InChI=1S/C12H20N4O6S/c17-11(14-8-3-5-13-6-4-8)10-2-1-9-7-15(10)12(18)16(9)22-23(19,20)21/h8-10,13H,1-7H2,(H,14,17)(H,19,20,21)/t9-,10+/m1/s1 | [InChIKey]
SMOBCLHAZXOKDQ-ZJUUUORDSA-N | [SMILES]
S(O)(ON1C(=O)[N@@]2C[C@@]1([H])CC[C@H]2C(NC1CCNCC1)=O)(=O)=O |
| Hazard Information | Back Directory | [Description]
Relebactam (1174018-99-5) is a novel,intravenous,class A and class C B-lactamase inhibitor and is currently under evaluation in combination with imipenem/cilastatin for the treatment of resistant Gram-negative infections.In vitro studies demonstrated that relebactam restored imipenem's activity against KPC-producing Enterobacteriacae�,lowering imipenem MICs from 16-64 to 0.12-1mg/L at a concentration of 4 mg/L. | [Uses]
Relebactam is a novel β-lactamase inhibitor in combination with Primaxin. | [Mechanism of action]
Relebactam is a beta-lactamase inhibitor known to inhibit many types of beta-lactamases including Ambler class A and Ambler class C enzymes, helping to prevent imipenem from degrading in the body.Label,Similar to the structurally-related avibactam, first, relebactam binds non-covalently to a beta-lactamase binding site, then, it covalently acylates the serine residue in the active site of the enzyme.In contrast to some other beta-lactamase inhibitors, once relebactam de-acylates from the active site, it can reform it's 5 membered ring and is capable of rebinding to target enzymes.
https://go.drugbank.com/drugs/DB12377 | [Clinical Use]
Recently, another carbapenem-β-lactamase inhibitor, imipenem/cilastatin-relebactam (RecarbrioTM), was approved by the FDA. Relebactam, a bicyclic diazabicyclooctane, is structurally related to avibactam but differs by the addition of a piperidine ring to the 2-position carbonyl group. Like meropenem- vaborbactam (VabomereTM), imipenem-relebactam(RecarbrioTM) is active against class A and class C carbapenemases and is approved for the treatment of multidrug-resistant intraabdominal infections secondary to Bacteroides caccae, Bacteroides fragilis, Bacteroides ovatus, Bacteroides thetaiotaomicron, Bacteroides uniformis, Bacteroides vulgatus, Bifidobacterium stercoris, Citrobacter freundii, Enterobacter cloacae, Escherichia coli, Fusobacterium nucleatum, Klebsiella aerogenes, Klebsiella oxytoca, Klebsiella pneumoniae, Parabacteroides distasonis, and Pseudomonas aeruginosa and multidrug-resistant complicated urinary tract infections secondary to E. cloacae, E. coli, K. aerogenes, K. pneumoniae and P. aeruginosa. | [Synthesis]
Relebactam(MK-7655) is a β-lactamase inhibitor in clinical trials as a combination therapy for the treatment of bacterial infection resistant to β-lactam antibiotics. Its unusual structural challenges have inspired a rapid synthesis featuring an iridium-catalyzed N–H insertion and a series of late stage transformations designed around the reactivity of the labile bicyclo[3.2.1]urea at the core of the target.
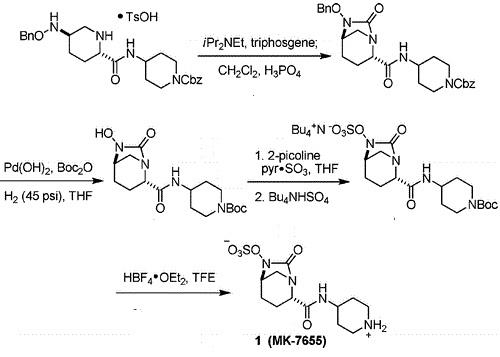
In 2014, Qualified Infectious Disease Product (QIDP) and Fast Track designations were assigned by the FDA for the treatment of complicated urinary tract infections, complicated intra-abdominal infections and hospital-acquired bacterial pneumonia/ventilator-associated bacterial pneumonia.
A Concise Synthesis of a β-Lactamase Inhibitor |
|
|